Is epoxidation syn or anti

Robert Spencer
Published Apr 10, 2026
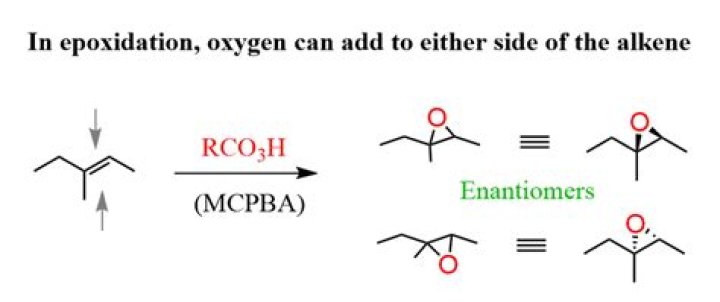
The epoxidation reaction is believed to occur in a single step with a transition state incorporating all of the bonding events shown in the equation. Consequently, epoxidations by peracids always have syn-stereoselectivity, and seldom give structural rearrangement.
Is Hydroboration a concerted reaction?
The hydroboration of alkenes is a single-step concerted mechanism. The π electrons act as the nucleophile with the electrophilic B atom, and the H is transferred to the C with syn stereochemistry. Everything happens at the same time.
Is epoxidation racemic?
The racemic epoxide is designated (2R*,3R*)-3-ethyl-2-methyloxirane. When (Z)-2-pentene undergoes the chlorohydrin reaction, there are likewise two possible racemic chlorohydrins formed, which upon base treatment, lead to a single, racemic epoxide. The formation of epoxides by the halohydrin route is stereospecific.
Is epoxidation anti addition?
The epoxide is first protonated followed by back-side attack by water. It is this back-side attack that is the ultimate reason that this reaction results in anti addition.Why is epoxidation syn addition?
Alkene epoxidation is an exciting reaction in which a double bond reacts with a peroxy acid to form an epoxide. This is considered a syn addition since the oxygen is bound to 2 carbon atoms.
What is epoxidation of alkenes?
Oxacyclopropane rings, also called epoxide rings, are useful reagents that may be opened by further reaction to form anti vicinal diols. One way to synthesize oxacyclopropane rings is through the reaction of an alkene with peroxycarboxylic acid.
Is epoxidation a stereospecific?
The epoxidation of cholesterol is stereoselective because the C-19 methyl group hinders attack from the top side of the molecule.
Is hydroboration an addition reaction?
In chemistry, hydroboration refers to the addition of a hydrogen-boron bond to C-C, C-N, and C-O double bonds, as well as C-C triple bonds. This chemical reaction is useful in the organic synthesis of organic compounds.What is borane made of?
Boranes are compounds composed solely of boron and hydrogen (and sometimes a cation) and may be neutral or anionic. History: The chemistry of the boranes was first studied systematically by Alfred Stock and his research group beginning in 1912 and continuing for almost 25 years.
Why is epoxidation useful?Epoxidation is a useful chemical reaction – the products of which have many uses in different markets, such as epoxidised soy oils as acid scavengers, making linear epoxides for derivatisation for composite materials, making 1,2-diols for personal care or complicated ring epoxides for F&F as well as intermediates for …
Article first time published onWhy is epoxidation of an alkene important?
Epoxides are an important class of compounds in organic synthesis, because nucleophilic ring opening takes place easily in an SN2 pathway with inversion of configuration at the reacting carbon center. The driving force of the high reactivity is the inherent strain of the three-membered heterocycle.
Is a Halohydrin an alcohol?
In organic chemistry a halohydrin (also a haloalcohol or β-halo alcohol) is a functional group in which a halogen and a hydroxyl are bonded to adjacent carbon atoms, which otherwise bear only hydrogen or hydrocarbyl groups (e.g. 2-chloroethanol, 3-chloropropane-1,2-diol).
Which reagent can be used for the epoxidation of the alkenes?
Oxacyclopropane rings, also called epoxide rings, are useful reagents that may be opened by further reaction to form anti vicinal diols. One way to synthesize oxacyclopropane rings is through the reaction of an alkene with a peroxycarboxylic acid, such as MCPBA (m-chloroperoxybenzoic acid).
What is asymmetric epoxidation?
The Asymmetric Epoxidation, or AE, involves the conversion of an allylic alcohol to an epoxy alcohol. Titanium (IV) isopropoxide is used as a catalyst and (+) or (-) diethyl or diisopropyl tartrate as a chiral ligand.
What is a peracid in organic chemistry?
peroxy acid, also called Peracid, any of a class of chemical compounds in which the atomic group ―O―O―H replaces the ―O―H group of an oxy acid (a compound in which a hydrogen atom is attached to an oxygen atom by a covalent bond that is easily broken, producing an anion and a hydrogen ion).
What is a syn addition?
Syn addition: An addition reaction in which all new bonds are formed on the same face of the reactant molecule.
What is meant by epoxidation?
Definition of epoxidation : a conversion of a usually unsaturated compound into an epoxide.
What is epoxidation Mcq?
Solution: Epoxidation is an example of electrophilic ad dition at olefin ic bonds. Therefore, greater the electron density at olefinic bond, greater its reactivity. … Solution: It gives racemic mixture of oxiranes.
What type of reaction is epoxidation?
Epoxidation is the chemical reaction which converts the carbon–carbon double bond into oxiranes (epoxides), using a variety of reagents including air oxidation, hypochlorous acid, hydrogen peroxide, and organic peracid (Fettes, 1964).
What is stereoselective and stereospecific?
A stereospecific mechanism specifies the stereochemical outcome of a given reactant, whereas a stereoselective reaction selects products from those made available by the same, non-specific mechanism acting on a given reactant.
What is regiospecific and stereospecific?
Stereoselective — the reaction can result in more than one stereoisomer but has some reason to prefer one over the other(s) (E2 dehydrohalogenation preferentially forms trans products) Regiospecific — the reaction can only result in one constitutional isomer (Markovnikov addition to an alkene)
What determines the rate of epoxidation?
The outcome of the epoxidation of a double bond is determined by the substituents; the effect of solvent polarity on the reaction rate is negligible. … Electron-donating substituents at the C=C double bond, for example alkyl groups, increase the rate of addition of the electrophilic oxygen atom.
Is Halohydrin formed by markovnikov?
Reaction Overview: The Halohydrin formation reaction involves breaking a pi bond and creating a halohydrin in its place. … This reaction takes place in water and yields an anti-addition reaction which follows Markovnikov’s rule.
What is the purpose of electrophilic addition?
Electrophilic addition reactions are an important class of reactions that allow the interconversion of C=C and C≡C into a range of important functional groups including alkyl halides and alcohols. Conceptually, addition is the reverse of elimination (see Chapter 5) which can be used to prepare alkenes.
Is Bh3 possible?
Bh3 molecule is electron deficient in nature since ‘B’ atom has only 6 electrons around it. So it exists in the form of dimer that is B2H6. Bf3 is also deficient but because of larger size of ‘F’ atom, it can’t undergo dimarization.
Does bh5 exist?
In a recent paper in Dalton Transactions, authors Szieberth, Szpisjak, Turczel and Konczol describe BH5 as “rare.” This is an understatement. As they report, its existence was confirmed in 1994 by infra-red spectroscopy in an argon matrix at 10-25K temperatures.
Is borane ionic or covalent?
The B−N dative bond in ammonia borane is 65% ionic, moderately strong (−27.5 ± 0.5 kcal/mol), and structurally flexible on the donor side to relieve steric congestion.
Is Hydroboration a Markovnikov addition?
Hydroboration–oxidation is an anti-Markovnikov reaction, with the hydroxyl group attaching to the less-substituted carbon. The reaction thus provides a more stereospecific and complementary regiochemical alternative to other hydration reactions such as acid-catalyzed addition and the oxymercuration–reduction process.
Is Hydroboration-oxidation a syn addition?
Hydroboration-oxidation is a two step pathway used to produce alcohols. … The hydroboration mechanism has the elements of both hydrogenation and electrophilic addition and it is a stereospecific (syn addition), meaning that the hydroboration takes place on the same face of the double bond, this leads cis stereochemistry.
Is Hydroboration racemic?
You get a racemic mixture of 2-ethylcylohexanol enantiomers. The overall reaction amounts to an “anti-Markovnikov” addition of H-OH to the C=C bond. Also, the reaction has syn stereochemistry, because the H-BH₂ adds to the same side of the alkene.
Are epoxides toxic?
Most epoxides are toxic because their high reactivity makes them mutagenic.