Is elemental sulfur acidic

Victoria Simmons
Published Apr 08, 2026
Elemental sulfur sources are highly acidifying. This can benefit your crops under alkaline soil conditions, but hurt them under acid conditions. Sulfate sources can be either acidifying or neutral in reaction.
Does elemental sulfur increase pH?
Elemental sulfur can be applied as a soil amendment to decrease the pH or acidify such soils. Due to the cost, the application of sulfur to acidify soils is more practical for horticultural crops than agronomic crops.
How much does sulfur lower pH?
Use about 4 to 6 lb. of aluminum sulfate per plant for most medium- and fine-textured Wisconsin soils in order to decrease soil pH by about one unit. If elemental sulfur is applied, decrease the total recommended application by one-sixth. One pound of aluminum sulfate or elemental sulfur is equal to about 2 cups.
Is Sulphur acidic or alkaline?
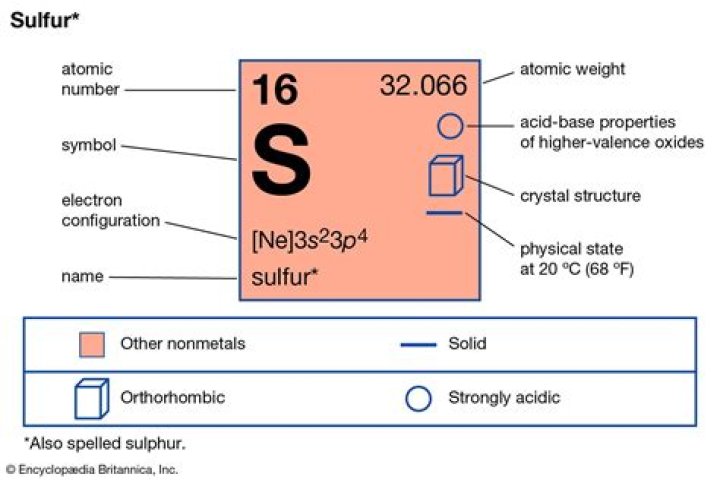
SulfurOxidation states−2, −1, 0, +1, +2, +3, +4, +5, +6 (a strongly acidic oxide)ElectronegativityPauling scale: 2.58Ionization energies1st: 999.6 kJ/mol 2nd: 2252 kJ/mol 3rd: 3357 kJ/mol (more)Covalent radius105±3 pmWhat is the pH of Sulphur dioxide?
The rate of solution of the gas was rapid for the first V2 hr during which time the pH decreased from 6.1 to approximately 4.0. As the rate of solution of atmospheric sulfur dioxide decreased the pH of the water decreased to a final value of 3.8.
How long does it take for sulfur to lower pH?
The soil acidification process depends on microorganisms’ activity and soil aeration, thus the reaction is slow in cold, wet soils. In normal conditions, it is expected that pH will decrease within three to six months after the application of elemental sulfur or ammonium-based fertilizers.
What is elemental sulfur used for?
In agricultural applications, ES can be used as an insecticide, an acaricide, a rodenticide, a repellent, a fertilizer, and a soil amendment to lower soil pH. How does elemental sulfur work as a pesticide? ES kills fungi on contact, likely by affecting fungal cell respiration.
Will lime lower pH in soil?
Lime is a soil amendment made from ground limestone rock, which naturally contains calcium carbonate and magnesium carbonate. When lime is added to soil, these compounds work to increase the soil’s pH, making soil less acidic and more alkaline.How much sulfur is needed to lower pH in lawn?
Some distributors and soil testing labs will recommend applying sulfur to reduce the soil pH of turf when it is over 7.5. Unfortunately, you can only apply 5 lbs sulfur/1000 sq. ft. per application to avoid burning turfgrass mowed at 2 inches or more, and a total of 10 lbs sulfur/1000 sq.
What is elemental sulphur fertilizer?The use of elemental S to reduce soil pH and to reclaim sodic soils is well known. However, its function as a fertilizer source of S has greatly increased its use in agriculture with the limited production and availability of other sulphur-containing fertilizer materials.
Article first time published onIs elemental sulfur organic?
Sulfur (Elemental 90%) OMRI Listed OMRI listed for certified organic production. 90% sulfur for soil application. Useful in lowering soil pH. Excellent for blueberries ,rhododendrons, azaleas and other acid loving plants.
Is sulphur trioxide acidic or basic?
Sulfur trioxide is an oxidant. It oxidizes sulfur dichloride to thionyl chloride. SO3 is a strong Lewis acid readily forming adducts with Lewis bases. With pyridine, it give the sulfur trioxide pyridine complex.
Does lime increase pH?
Adding lime (Figure 1) increases soil pH (reduces acidity), adds calcium (Ca) and/or magnesium (Mg), and reduces the solubility of Al and Mn in the soil. aWhen soil pH is below the minimum value, crop yields may be reduced.
How long does elemental sulfur last in soil?
Elemental sulfur can take one to two years to convert into sulfuric acid and lower the pH of the soil.
How do I apply elemental sulfur to my lawn?
In the spring, apply five pounds of sulfur per 1,000 square feet of grass and water it into the existing lawn. For bare spots, spread sulfur and then mix it into the top six inches of soil.
What is the pH of Na?
BaseName100 mMSr(OH)2strontium hydroxide13.09NaOHsodium hydroxide12.88KOHpotassium hydroxide (caustic potash)12.88Na2SiO3sodium metasilicate12.62
What is sulfur dioxide made of?
Sulfur dioxide, SO2, is a colorless gas or liquid with a strong, choking odor. It is produced from the burning of fossil fuels (coal and oil) and the smelting of mineral ores (aluminum, copper, zinc, lead, and iron) that contain sulfur.
Why is SO2 acidic in nature?
YOUR ANSWER IS HERE. When sulfur dioxide is dissolved in rain, it makes sulfurous acid in the reaction SO2 + H2O -> H2SO3. It is because of a very similar reason why CO2 is just a bit acidic, and that is because CO2 + H2O (maybe in the form of rain) -> H2CO3.
Is Elemental sulphur reactive?
sulfur (S), also spelled sulphur, nonmetallic chemical element belonging to the oxygen group (Group 16 [VIa] of the periodic table), one of the most reactive of the elements. … It reacts with all metals except gold and platinum, forming sulfides; it also forms compounds with several nonmetallic elements.
Will gypsum raise soil pH?
Gypsum does not change pH nor improve drainage in non-sodic situations. Gypsum is used to add calcium to soils such as serpentine with very high or toxic Mg levels.
Will coffee grounds acidify soil?
Conclusion: Coffee grounds are a good addition to the compost pile. … It is doubtful that they have a great effect on pests, but unless the grounds are very acidic, they will NOT acidify your soil.
Can you use vinegar to lower pH in soil?
To lower the pH level of soil and make it more acidic, vinegar can be applied by hand or using an irrigation system. For a basic treatment, a cup of vinegar can be mixed with a gallon of water and poured over soil with a watering can.
Does Ironite lower pH?
As Fertilizer Ironite is marketed as a soil amendment for alkaline soils. … Ironite is thus used to adjust the soil’s pH to make it less alkaline and to allow the plant to more easily absorb iron and phosphorus.
What pH is lime soil?
Should I lime my soil to pH 6.0, 6.5 or 6.8? For most crops, liming to pH 6.5 is recommended. When alfalfa is grown, liming to pH 6.8 is essential. For gardens, potatoes or low-maintenance grass pasture, liming to pH 6.0 will be satisfactory.
When should I apply Sulphur to my lawn?
Because an abundance of sulfur can burn the grass, you must apply it over time. You can apply up to 5 pounds of elemental sulfur per 1,000 square feet of an established lawn. Make applications every three to four weeks during the cooler fall and spring weather.
What should the pH of a lawn be?
The ideal pH range is between 6.0 and 7.0 – anything higher or lower may result in unhealthy turf. Your lawn is considered acidic when the pH of the soil falls below 6.0, at which point a lime treatment would be beneficial.
What fertilizer will lower pH of soil?
As previously mentioned, if a lower pH is required, ammonium or urea-based fertilizers can aide in the reduction of pH.
What is the pH of lemon?
Lemon juice has a pH between 2 and 3, which means it’s 10,000–100,000 times more acidic than water (1, 2, 3). A food’s pH is a measure of its acidity. The pH of lemon juice falls between 2 and 3, meaning it is acidic.
What do farmers Add to reduce the acidity of soil?
Limestone acts as a soil acid neutralizer and consists of either calcium and magnesium carbonate or calcium carbonate. These are called dolomitic limestone and calcitic limestone respectively.
Is vinegar acidic or alkaline?
Vinegar is acidic. Vinegar’s pH level varies based upon the type of vinegar it is. White distilled vinegar, the kind best suited for household cleaning, typically has a pH of around 2.5. Vinegar, which means “sour wine” in French, can be made from anything containing sugar, such as fruit.
Is Elemental Sulphur water soluble?
The characteristics of elemental sulphur are: non-metallic chemical element; a yellow crystalline solid; insoluble in water; odorless; and 99.5% pure. Elemental sulphur is utilized in several industries other than agriculture.



