Is diamond a network solid

Rachel Hickman
Published Apr 10, 2026
In a network solid there are no individual molecules, and the entire crystal or amorphous solid may be considered a macromolecule. … Examples of network solids include diamond with a continuous network of carbon atoms and silicon dioxide or quartz with a continuous three-dimensional network of SiO2 units.
Is diamond a metallic solid?
Characterized as being very hard with very high melting points and being poor conductors. Examples of this type of solid are diamond and graphite, and the fullerenes. … Metallic solids—Made up of metal atoms that are held together by metallic bonds.
Which is the network solid?
The correct answer is (B) Diamond. Diamond is called a network solid. Any chemical compound in which covalent bonds bond the atoms in a continuous network extending throughout the material is called network solid, or covalent network solid or atomic crystalline solids.
What type of network solid is diamond?
Covalent Network Solids are giant covalent substances like diamond, graphite and silicon dioxide (silicon(IV) oxide).Is diamond a metal?
Diamond is not considered as a non-metal in the exceptional category as diamond is a form of carbon. It is not classified as an element. … It is an allotrope of carbon.
Is diamond a covalent?
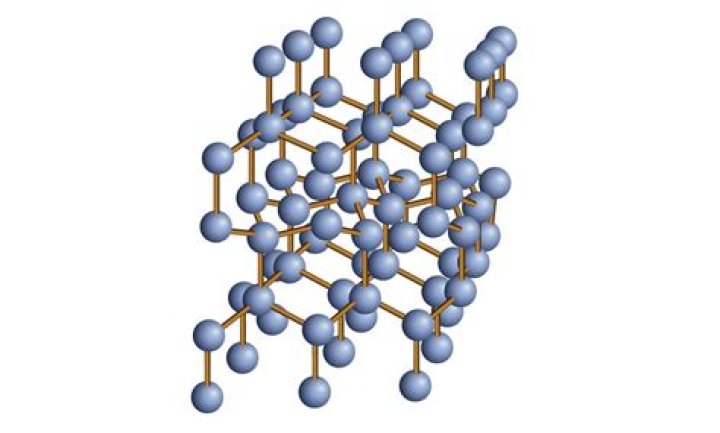
Diamond is a giant covalent structure in which: each carbon atom is joined to four other carbon atoms by strong covalent bonds. the carbon atoms form a regular tetrahedral network structure.
Is diamond an amorphous solid?
Some examples include sodium chloride, ice, metals, and diamonds. Amorphous solids, on the other hand, are rigid, but they lack repeated periodicity or long-range order in their structure. … Amorphous solids include both natural and man-made materials. The most frequently cited example of an amorphous solid is glass.
Are diamonds molecular?
Diamond is a covalent network solid, like a number of other common materials (quartz, graphite, glass, and a whole bunch of stuff). Because they are not discrete molecules – there is no ‘diamond’ molecule the same way there are molecules of caffeine, benzoic acid, citric acid, N,N-dimethylaminopyridine, etc.What is a metallic solid?
Metallic solids are composed of metal cations held together by a delocalized “sea” of valence electrons. … Metallic solids also tend to be malleable and ductile due to the ability of the metal nuclei to move past each other without disrupting the bonding.
Is diamond a compound?The quick answer is: Diamond is a pure element, carbon; gold is a pure element, gold; and rust is a compound, Iron Oxide, of iron and Oxygen. Diamond is pure elemental carbon, compressed to its crystal form, under extreme heat and pressure deep within the Earth.
Article first time published onWhat is a diamond an example of?
Hence, diamond is an example of network covalent solid.
Why is diamond a covalent network solid?
Covalent Network Solids. Covalent solids are formed by networks or chains of atoms or molecules held together by covalent bonds. … (a) Diamond consists of sp3 hybridized carbon atoms, each bonded to four other carbon atoms. The tetrahedral array forms a giant network in which carbon atoms form six-membered rings.
Which is not a network solid?
– Solid sulfur dioxide is a molecule and so even though it is a solid and has covalent bonding, it is not a network solid because it is a molecule.
Which of the following is a network solid SO2 I2 diamond H2O?
Which of the following is a network solid? (a) SO2 (solid) (b) I2 (c) Diamond (d) H2O (ice) Answer: (c) Diamond is a giant molecule in which constituent atoms are held together by covalent bond. Hence, this is a network solid.
Is diamond a conductor?
Normally an insulator, diamond becomes a metallic conductor when subjected to large strain in a new theoretical model. … Long known as the hardest of all natural materials, diamonds are also exceptional thermal conductors and electrical insulators.
Why diamond is non metallic?
Diamond is one form of carbon which is formed at extreme thermodynamic conditions. And well as we all know that Diamond is actually the hardest natural substance on Earth. So, diamond is essentially non-metal.
Is diamond a stone or a metal?
diamond, a mineral composed of pure carbon. It is the hardest naturally occurring substance known; it is also the most popular gemstone. Because of their extreme hardness, diamonds have a number of important industrial applications.
Is diamond non crystalline or amorphous?
Diamond is crystalline and anisotropic, meaning that its properties are directional. The single crystalline diamond shown in the left picture contains lots of facets. In contrast, amorphous diamond is isotropic like glass, and it may be cut to any shape including an ideal sphere.
What are examples of amorphous solids?
amorphous solid, any noncrystalline solid in which the atoms and molecules are not organized in a definite lattice pattern. Such solids include glass, plastic, and gel.
What is solid classification?
Solids are broadly classified into two types crystalline solids and amorphous solids. A crystalline solid is a substance whose constituent particles possess regular orderly arrangement e.g. Sodium chloride, sucrose, diamond etc.
Is diamond a graphite?
Mineral NameGraphiteDiamondCrystal SystemHexagonalIsometricCrystal ClassSpace GroupC63/mmcFd3m
Are diamond and graphite allotropes?
Diamond, graphite and fullerenes (substances that include nanotubes and ‘buckyballs’ , such as buckminsterfullerene) are three allotropes of pure carbon.
Is diamond a 3d structure?
Because the diamond structure forms a distance-preserving subset of the four-dimensional integer lattice, it is a partial cube. Yet another coordinatization of the diamond cubic involves the removal of some of the edges from a three-dimensional grid graph.
What are the examples of metallic?
- AgNO3 – Silver nitrate is a metallic compound. Silver (Ag) is the metal, bonded to the nitrate group.
- CaCl2 – Calcium chloride is a metallic compound.
- H2O (water) is not considered a metallic compound. Even though hydrogen sometimes acts like a metal, it is more often considered a nonmetal.
Which of the following are metallic solids?
Examples of metallic solids – copper, gold, zinc etc. Note: We generally think metallic solids as pure metals, but they can also be the combinations of metals that are alloys like bronze which is a mixture of copper and tin.
What are covalent or network solids?
Covalent network solids are composed of atoms covalently bonded together into a three-dimensional network or layers of two-dimensional networks. Due to the strength of the covalent bonds, covalent network solids have high melting points.
Why is a diamond a solid?
Diamond is a solid form of the element carbon with its atoms arranged in a crystal structure called diamond cubic. At room temperature and pressure, another solid form of carbon known as graphite is the chemically stable form of carbon, but diamond almost never converts to it.
Is diamond a molecule or element?
Diamond is composed of the single element carbon, and it is the arrangement of the C atoms in the lattice that give diamond its amazing properties. Compare the structure of diamond and graphite, both composed of just carbon.
Is diamond a molecule or atom?
A diamond is one giant molecule of carbon atoms. Diamonds are colourless and transparent . They sparkle and reflect light, which is why they are described as lustrous .
Is diamond a mixture or element or compound?
It’s a heterogeneous mixture. Diamond is made of just one element: carbon. Each carbon atom in diamond is connected to four other carbon atoms, in a crystal that extends on and on. There are other forms of pure carbon where the atoms are bonded differently, notably charcoal and graphite.
What type of compound is diamond?
Diamond has a giant covalent structure in which: each carbon atom is joined to four other carbon atoms by covalent bonds. the carbon atoms have a regular lattice arrangement. there are no free electrons.



