Is cyclohexanol alcoholic

Robert Spencer
Published Feb 14, 2026
Cyclohexanol is an alcohol that consists of cyclohexane bearing a single hydroxy substituent. The parent of the class of cyclohexanols. It has a role as a solvent. It is a secondary alcohol and a member of cyclohexanols.
Why is cyclohexanol a alcohol?
Cyclohexanol, C6H12 , is formed by the oxidation of cyclohexane, C6H11OH . In the formation of cyclohexanol, one hydrogen atom on the cyclohexane ring is replaced by a hydroxyl group (-OH), which makes it an alcohol.
Can you drink cyclohexanol?
* Do not eat, smoke, or drink where Cyclohexanol is handled, processed, or stored, since the chemical can be swallowed.
Is cyclohexanol cyclic alcohol?
SynonymSourceCyclohexan-1-olChEBICyclohexanolsMeSHCyclohexyl alcoholChEBIHexahydrophenolChEBIWhat type of alcohol is Cyclopentanol?
Cyclopentanol or cyclopentyl alcohol is a cyclic alcohol. It is also known as hydroxycyclopentane.
Are cyclohexane and cyclohexanol the same?
is that cyclohexane is (organic compound) an alicyclic hydrocarbon, c6h12, consisting of a ring of six carbon atoms; a volatile liquid while cyclohexanol is (organic compound) the alicyclic alcohol derived from cyclohexane by replacement of a hydrogen atom with a hydroxyl group.
Why is cyclohexanol a secondary alcohol?
Each carbon atom is bonded to the adjacent two carbon atoms. … In cyclohexanol, the hydroxyl $\text{ }-\text{OH }$group is attached to one of the carbon atoms of the cyclohexane. Thus, the carbon atom bearing a hydroxyl group is secondary carbon. Thus, cyclohexanol is a secondary alcohol.
Which of the following is aromatic alcohol?
The IUPAC name of this compound is phenyl methanol. This compound consists of a hydroxyl group attached to a methyl group, which is in turn attached to an aromatic ring. This structure satisfies the requirements to be aromatic alcohol.Is cyclohexanol aliphatic or aromatic?
Definition : An aliphatic compound having a carbocyclic ring structure which may be saturated or unsaturated, but may not be a benzenoid or other aromatic system.
Which is the secondary alcohol?A secondary alcohol is a compound in which a hydroxy group, ‒OH, is attached to a saturated carbon atom which has two other carbon atoms attached to it.
Article first time published onIs ethyl benzoate harmful?
Inhalation May be harmful if inhaled. May cause respiratory tract irritation. Ingestion May be harmful if swallowed. Skin May be harmful if absorbed through skin.
Is sodium carbonate hazardous?
Hazards Associated with Sodium Carbonate If improperly handled, or in the event of accidental exposure, sodium carbonate can pose several hazards to your health and safety. Inhalation of this chemical can lead to adverse effects such as respiratory tract irritation, coughing, shortness of breath, and pulmonary edema.
How do you convert cyclohexanol to cyclohexene?
One molecule of cyclohexanol should produce one molecule of cyclohexene. One mole (mol) of cyclohexanol should produce one mole of cyclohexene. If 2.05 g of cyclohexanol is used (use the actual amount used in your experiment) convert this to moles by dividing by the molecular weight of cyclohexanol (MW = 100.2 g/mol).
Is cyclohexanol toxic?
Cyclohexanol appears as a colorless liquid with a camphor-like odor. Soluble in most organic liquids. Flash point 154°F. May be toxic by inhalation or skin exposure.
Is Cyclopentanol primary alcohol?
Cyclopentanol, 99%, Thermo Scientific™: Secondary alcohols Alcohols and polyols | Fisher Scientific.
What does Cyclopentanol look like?
Cyclopentanol appears as a colorless viscous liquid with a pleasant odor. Slightly less dense than water. … Cyclopentanol is the simplest member of the class of cyclopentanols bearing a single hydroxy substituent. The parent of the class of cyclopentanols.
What is the common name of cyclohexanol?
NamesOther names Cyclohexyl Alcohol, hexahydrophenol, hydrophenol, hydroxycyclohexane, Naxol Hexalin Hydralin HOCyIdentifiersCAS Number108-93-03D model (JSmol)Interactive image
Is cyclohexanol an acid or base?
Cyclohexanol has a pKa of about 18. It is less acidic than water. It can give up a proton, but the proton is much more likely to be bound to the oxygen than disscociated. Phenol has a pKa of about 9 and is considered mildly acidic.
Can cyclohexanol be oxidized?
When cyclohexanol is exposed to sodium hypochlorite (NaOCl) and acetic acid, an oxidation reaction takes place that gives cyclohexanone as the product.
Is benzyl alcohol a primary alcohol?
Benzyl Alcohol, CAS No. 100-51-6 is an aromatic primary alcohol with the molecular formula C7H8O. It is a clear, oily liquid with a mild, pleasant odor. It is soluble in water and readily soluble in alcohol and ether.
How many hydrogens are in cyclohexanol?
Thus, this structure has a total of six carbon atoms, twelve hydrogen atoms, and one oxygen atom. To get started with physical properties, we should know the following about cyclohexanol: It has a melting point of 25.93 degrees Celsius. It has a boiling point of 161.84 degrees Celsius.
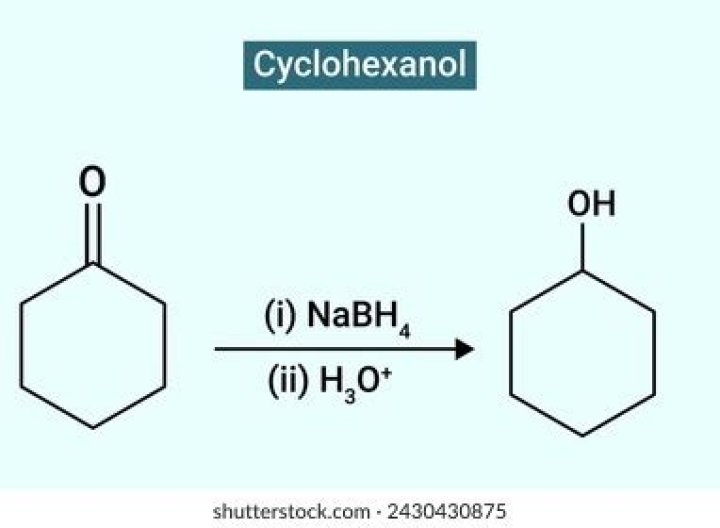
How do you make cyclohexanol?
Procedure: Dissolve sodium dichromate dihydrate (12.5 g) in water (60 mL) in a 100 mL beaker and with continuous stirring with a glass rod (NOTE), carefully and slowly add concentrated sulfuric acid (11 g, 6 mL). Allow the mixture to cool.
What functional group does cyclohexanol have?
Cyclohexanone is the organic compound with the formula (CH2)5CO. The molecule consists of six-carbon cyclic molecule with a ketone functional group.
Is cyclohexanol polar?
I would classify cyclohexanol as mostly non polar, as the ring is relatively bulky. At the same time there is no doubt it is much more polar than cyclohexane.
Which of the following is not aromatic alcohol?
Phenol is also known as, carbolic acid’ cannot be considered as aromatic alcohol. It is quite separate branch of compound called phenols.
Is phenol an alcohol?
Phenols have unique properties and are not classified as alcohols. They have higher acidities due to the aromatic ring’s tight coupling with the oxygen and a relatively loose bond between the oxygen and hydrogen.
What is the difference between phenol and aromatic alcohol?
Difference Between Alcohol and PhenolAlcohols show no impact or reaction during tests as they are mostly neutral.Phenol can change litmus paper red as they are acidic in nature.
How do you tell if an alcohol is primary or secondary?
Alcohols are organic molecules containing a hydroxyl functional group connected to an alkyl or aryl group (ROH). If the hydroxyl carbon only has a single R group, it is known as primary alcohol. If it has two R groups, it is a secondary alcohol, and if it has three R groups, it is a tertiary alcohol.
What are the 3 types of alcohol?
In chemistry, an alcohol exists when a hydroxyl group, a pair of oxygen and hydrogen atoms, replaces the hydrogen atom in a hydrocarbon. Alcohols bind with other atoms to create secondary alcohols. These secondary alcohols are the three types of alcohol that humans use every day: methanol, isopropanol, and ethanol.
What are the 4 types of alcohol?
The four types of alcohol are ethyl, denatured, isopropyl and rubbing. The one that we know and love the best is ethyl alcohol, also called ethanol or grain alcohol. It’s made by fermenting sugar and yeast, and is used in beer, wine, and liquor.
Is ethyl benzoate aromatic?
Namesshow SMILESPropertiesChemical formulaC9H10O2Molar mass150.177 g·mol−1