Is cinnamic acid a solvent

Christopher Lucas
Published Apr 10, 2026
NamesHazard statementsH315 , H319 , H335
Is cinnamic acid soluble in dichloromethane?
The cinnamic acid is soluble in dichloromethane at room temperature and thus before the bromine addition the reaction vessel holds a colourless solution. … As the cinnamic acid is soluble in cold CH2Cl2 the washing of the final product is essential to assure a good purity.
Is cinnamic acid soluble in hot water?
Properties of Cinnamic AcidNameCinnamic AcidSolubility in WaterSlightly soluble
What is the solubility of benzoic acid?
NamesBoiling point250 °C (482 °F; 523 K)Solubility in water1.7 g/L (0 °C)2.7 g/L (18 °C) 3.44 g/L (25 °C) 5.51 g/L (40 °C) 21.45 g/L (75 °C) 56.31 g/L (100 °C)SolubilitySoluble in acetone, benzene, CCl4, CHCl3, alcohol, ethyl ether, hexane, phenyls, liquid ammonia, acetatesWhat element is present in cinnamic acid?
Cinnamic acid is a monocarboxylic acid that consists of acrylic acid bearing a phenyl substituent at the 3-position. It is found in Cinnamomum cassia. It has a role as a plant metabolite. It is a member of styrenes and a member of cinnamic acids.
Is cinnamic acid an alkene?
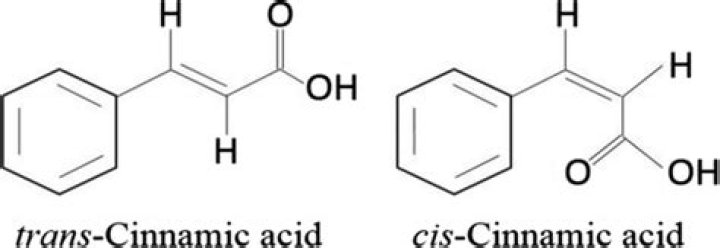
In contrast, trans -cinnamic acid (shown below) is a more complicated alkene that is found in a variety of natural sources including oil of cinnamon. Bromination of trans -cinnamic acid can result in the formation of two different pairs of enantiomers (see Figure 4).
Is M toluic acid soluble in water?
Chemical Properties: white or yellow crystal, almost insoluble in water, slightly soluble in boiling water, soluble in ethanol, ether.
What is cinnamic acid derivatives?
The common cinnamic acid derivatives include p-coumaric acid, caffeic acid, and ferulic acid. Chokeberry is abundant in hydroxycinnamic acid derivatives represented mainly by chlorogenic acid and neochlorogenic acid. … Hydroxybenzoic acid is the most abundant phenolic acid in cranberry followed by hydroxycinnamic acid.What is the use of cinnamic acid?
trans-Cinnamic acid is used in the manufacture of flavors, dyes, and pharmaceuticals; but its major use is for the production of its methyl, ethyl, and benzyl esters. These esters are important components of perfumes. The acid is also a precursor to the sweetener aspartame.
Is benzoic acid soluble in acetone?As can be seen from the figure, acetone exhibits the highest solubility for benzoic acid but very close to 2-propanol. At most temperatures, the solubility order in these solvents was determined as acetone > 2-propanol > acetic acid > cyclohexane.
Article first time published onIs benzoic acid soluble in MTBE?
The partition coefficient was calculated to be 0.961, indicating that the benzoic acid may be less soluble in organic than the water layer. … The partition coefficient is more commonly found to be 1.21.5 for benzoic acid in 1 mL MTBE and 1 mL water.
Is benzoic acid soluble in acid?
Benzoic acid or benzene-carbonic-acid is a monobasic aromatic acid, moderately strong, white crystalline powder, very soluble in alcohol, ether, and benzene, but poorly soluble in water (0.3 g of benzoic acid in 100 g of water at 20 °C).
Is cinnamic acid toxic?
Cinnamic acid is a compound of low toxicity, but its molecular structure and the known toxicity of similar molecules, such as styrene, have brought it to the toxicologist’s attention. … The related aldehyde, alcohol and esters are all more toxic than cinnamic acid.
Is salicylic acid soluble in water?
Salicylic acid is not readily soluble in water at room temperature. It needs to be converted to salicylate if you want it to dissolve completely in water. Its solubility is also dependent on the pH of the solvent. It dissolves very well in water at a pH level of 3.
Is cinnamic acid safe?
Inhalation – May cause respiratory irritation. Inhalation May be harmful if inhaled. Causes respiratory tract irritation. Ingestion May be harmful if swallowed.
Is cinnamic acid aromatic compound?
trans-Cinnamic acid, also known as (e)-cinnamic acid or phenylacrylic acid, belongs to the class of organic compounds known as cinnamic acids. These are organic aromatic compounds containing a benzene and a carboxylic acid group forming 3-phenylprop-2-enoic acid.
How is cinnamic acid made?
Cinnamic acid was synthesized using Perkin reaction by reacting 0.05 mole of benzaldehyde with 0.073 mole of acetic acid anhydride and 0.03 mole of sodium acetate as a catalyst in the Erlenmeyer flask and then the mixture was put in a sonicator for 60 minutes at 70 oC.
What are the reactants required to produce cinnamic acid?
Perkin reactionAromatic aldehyde + Aliphatic Acid anhydride + Alkali salt of the acid ↓ Cinnamic acid derivativesIdentifiersRSC ontology IDRXNO:0000003
Is M toluic acid polar or nonpolar?
MoleculeRelative Polarity and Commentstoluenenon-polarm-xylenenon-polar
Is toluic acid polar?
In p-toluic acid, it contains double-bonded oxygen and a single bonded one but it also contains a methyl group that is of almost the same electro-negativity as C in benzene. So, p-toluic acid is more polar than benzophenone. Therefore, p-toluic acid is the most polar followed by benzophenone and benzil.
Why is P-toluic acid not soluble in water?
ChEBI Namep-toluic acidChEBI ASCII Namep-toluic acidDefinitionA methylbenzoic acid in which the methyl substituent is located at position 4.
Is cinnamic acid soluble in ethyl acetate?
It is colorless to white prismatic crystals with the relative density being 1.284 (4 ℃), the melting point being 42 ℃, the boiling point 265 ℃ (decomposition ) and 125 ℃ (2.533 × 103Pa); it is slightly soluble in water (25 ℃ when 0.937) but easily soluble in alcohol, ether and ethyl acetate.
Is cinnamic acid saturated or unsaturated?
Cinnamic acid, an unsaturated carboxylic acid, is the chief constituent of the fragrant balsamic resin storax.
How is cinnamic acid formed by using Perkin's reaction?
The Perkin reaction is an organic reaction developed by William Henry Perkin that is used to make cinnamic acids. It gives an α,β-unsaturated aromatic acid by the aldol condensation of an aromatic aldehyde and an acid anhydride, in the presence of an alkali salt of the acid.
Where is cinnamic acid found?
Cinnamic acid is naturally found in the spice cinnamon, which is derived from the bark of trees from the genus Cinnamomum. The main aromatic compound responsible for the flavor of cinnamon is cinnamyl aldehyde.
Which building block is used for synthesis of cinnamic acid?
Cinnamic acids have been prepared in moderate to high yields by a new direct synthesis using aromatic aldehydes and aliphatic carboxylic acids, in the presence of boron tribromide as reagent, 4-dimethylaminopyridine (4-DMAP) and pyridine (Py) as bases and N-methyl-2-pyrolidinone (NMP) as solvent, at reflux (180-190°C) …
How cinnamic acid is synthesized from benzaldehyde?
Cinnamic acid was prepared by oxidation of benzalacetone which was synthesized by condensing benzaldehyde and acetone. For producing benzalacetone, benzaldehyde was reacted with acetone in 1% NaOH solution. … The benzalacetone was further oxidized with sodium hypochlorite to give 88.2% of cinnamic acid, m.p. 133°C.
Is acetone soluble or insoluble?
NamesSolubility in waterMiscibleSolubilityMiscible in benzene, diethyl ether, methanol, chloroform, ethanollog P−0.16
Is benzoic acid soluble in acetone at room temperature?
The experimental results showed that acetone had the best solubility for benzoic acid followed by 2-propanol, acetic acid and cyclohexane and the solubility in acetic acid had the strongest positive temperature dependency. The newly measured solubility data was also compared with the available literature data.
Does acetone react with benzoic acid?
Transcribed image text: Acetone reacts with Benzoic Acid to form a condensation product.
Is benzoic acid more soluble in water or MTBE?
more soluble in water than the unreacted benzoic acid. To force the benzoic acid to crystallize out of the aqueous layer, we added a small amount of HCl. … Wily t i vay onece po) Our extraction used MTBE (methyl tert-butyl ether) as our organic solvent and water.