Is be2 stable or unstable

Sophia Edwards
Published Feb 14, 2026
The electronic configuration of Beryllium is 1s^2 2s^2. From the electronic configuration it is clear that there is no singly filled atomic orbital
Is Be2 or H2 more stable?
Be2 is more stable because it contains both bonding and antibonding valence electrons.
What is the bond order of Be2?
The bond order for Be2 is 0 (zero). The valence shell of each beryllium atom is 2s2 so there are a total of four valence shell electrons for which we…
Is He2 a stable molecule?
Therefore, He2 molecules are not predicted to be stable (and are not stable) in the lowest energy ground state. However, in a high-energy environment, electron(s) from the antibonding orbitals in He2 can be promoted into higher-energy bonding orbitals, thus giving a nonzero bond order and a “reason” to form.Why Li2 is stable and Be2 is unstable?
Nb= Number of electrons in bonding orbitals Na= Number of electrons in anti-bonding orbitals According to Molecular Orbital Theory If Nb > Na, the molecule is stable If Nb=Na, the molecule is unstable If Nb<Na, the molecule is unstable The Molecular Orbital Configuration of Li2 is [σ1s]^2 [σ*1s]^2 [σ2s]^2 Bond Order = …
Is Ne2 stable or unstable?
With the help of molecular orbital theory show that Ne2 cannot exist as stable species .
Why is H2 more stable than Be2?
Be2 is more stable because it contains both bonding and antibonding valence electrons. II. H2 has a higher bond order than Be2. … H2 is more stable because it is diamagnetic, whereas Be2 is paramagnetic.
Is Be2 possible?
From the electronic configuration it is clear that there is no singly filled atomic orbital present in beryllium. Without the half- filled orbital, the overlapping is not possible, therefore Be2 molecule does not exist.Is He2 or He2+ more stable?
Molecular ion (He2)+ has a bond order of 0.5 , while (H2)+ has a bond order 0.5. Antibonding orbital is destabilized more in energy than bonding orbital stabilized. Now you should be able to answer the question. He2+ is the more stable of the pair because it has two electrons that it can release to form the ion.
Is He2 theory stable Mo?Re: Why is He2 not a stable molecule? [ENDORSED] The σ1s bonding and antibonding orbitals will be full. Calculating the bond order results in 0. In other words, no bond can be sustained between two He atoms according to MO theory.
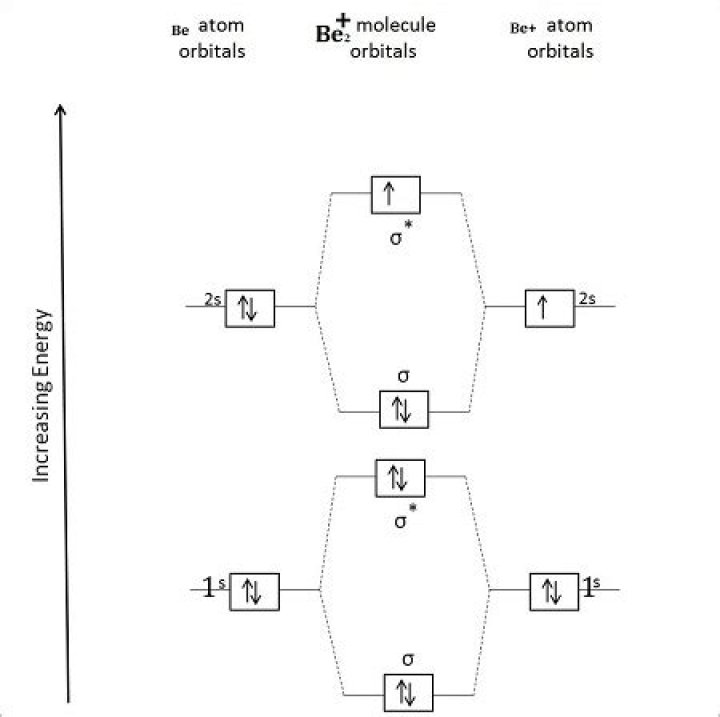
Article first time published onWhich is more stable Be2 Be2+?
Answer. Be2- is more stable than Be2, and Be2 is more stable thanBe2+. Be2 is more stable than either Be2+ or Be2-.
Is Be2 − Be2 − paramagnetic or diamagnetic?
Your question is wrong because be2 molecule does not exist as be2 number of electron is 8 so according to MOT its bond order comes out to be zero. Hence be2 is neither diamagnetic nor paramagnetic as it does not exist.
How many molecular orbitals are in Be2?
Therefore, \[\text{B}{{\text{e}}_{\text{2}}}\] molecule has eight electrons which are to be filled in four molecular orbitals. – Zero value of bond order corresponds to non-existence of \[\text{B}{{\text{e}}_{\text{2}}}\] molecule.
Does Be2 exist in gas phase?
Since both have a bond order of +12, both Be+2 and Be−2 will exist in gas phase.
Is C2 +2 stable?
According to the molecular orbital diagram of the (C2)2+ ion you its a stable ion, because it has a bond order of 1 & that means its a stable substance.
What is the equilibrium bond length?
equilibrium bond length: The average distance between two atoms when they are bonded to each other. … It is experimentally determined by measuring the heat (or enthalpy) required to break a mole of molecules into their constituent individual atoms.
Is Li2 more stable than Li2?
Li2 is more stable than Li+2 , because the bond is (hypothetically) stronger (probably gas-phase). … for each electron in an antibonding MO, it subtracts 0.5 from the bond order, because more antibonding character weakens the bond…
Why Ne2 is unstable?
The outermost shell in Ne element is fully filled with electrons where the electron is 10 in number. This means that will have 20 electrons. This is why it does not need to react with any other atom and so has no bond order and cannot exist.
Is Neon a Ne2 gas?
According to what I could understand from your question: Neon is a noble element which remains in gaseous state. Thus, Neon(Ne) itself is the gas. Neon is a chemical element with symbol Ne and atomic number 10.
Is N2+ stable or unstable?
Thus N2+ is more stable than N2– What is the bond order of N+2?
Why is Dihelium far less likely to exist than dihydrogen?
Dihydrogen (H2) with an Electron in the Antibonding Orbital The electron configuration of dihelium: If the molecule He2 were to exist, the 4s electrons would have to fully occupy both the bonding and antibonding levels, giving a bond order of zero. Dihelium does not exist.
When molecule or ion is stable?
Atoms are at their most stable when their outermost energy level is either empty of electrons or filled with electrons.
Is He2+ paramagnetic or diamagnetic?
Let it be H2- ,H2+ or He2+ ; there’s an unpaired electron i.e, they are paramagnetic.
Why be2 is not formed?
Be has electronic configuration 1S2,2S2 and s orbital is completely filled and doesn’t require any electron to complete its orbital. Hence, Be2 doesn’t exist at all.
Why is N2 more stable than N2?
N2 has highest bond order of 3 compared to N2+ which is 2.5, hence N2 it is more stable than N2+. Hope it helps you!
What is the HCH bond angle in ethane?
Ethane, C2H6, has a geometry related to that of methane. The two carbons are bonded together, and each is bonded to three hydrogens. Each H-C-H angle is 109.5° and each H-C-C angle is 109.5°.
How do you know if a molecule is stable?
- The greater the number of covalent bonds, the greater the stability since more atoms will have complete octets.
- The structure with the least number of formal charges is more stable.
- The structure with the least separation of formal charge is more stable.
Which is not a stable molecule?
The two oxygen atoms in this molecule follow the octet rule. Nitrogen dioxideNitrogen dioxide is another stable molecule that disobeys the octet rule. Note the seven electrons around nitrogen. Formal charges and the molecule’s resonance structures are indicated.
Is helium molecule stable?
Helium’s staunch stability is due to its closed-shell electronic configuration – its outer shell is complete, which means there’s no room for it to bond with other atoms by sharing electrons.
Is F2 a bond order?
Therefore F2+ will have the highest bond order and strongest bond F2- will have the weakest bond and therefore the longest bond. Both F2+ and F2- will have an odd number of electrons and thus 1 unpaired electron.
Does be2 2 have unpaired electrons?
Magnetic property: Since bond order is zero, Be2 molecule does not exist. It is diamagnetic due to the absence of any unpaired electron.



