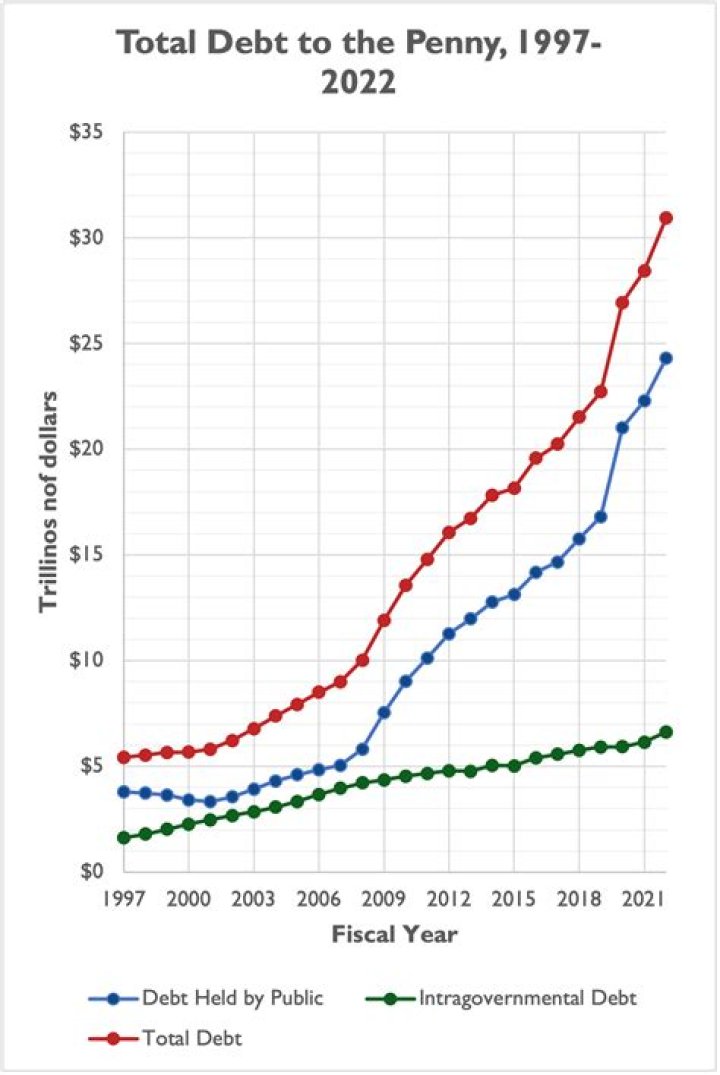
Is barium nitride aqueous

Sophia Edwards
Published Feb 26, 2026
NamesSolubility in water4.95 g/100 mL (0 °C) 10.5 g/100 mL (25 °C) 34.4 g/100 mL (100 °C)Solubilityinsoluble in alcohol
Is barium nitride a solid liquid or gas?
Compound FormulaBa3N2Molecular Weight439.994AppearanceOrange-yellow solid or powderMelting PointN/ABoiling PointN/A
Is barium hydroxide soluble?
Calcium hydroxide solution is referred to as “lime water”. A liter of pure water will dissolve about 1 gram of calcium hydroxide at room temperature. Barium hydroxide is soluble enough to produce a solution with a concentration around 0.1 mol dm-3 at room temperature.
Is barium sulfate soluble or insoluble?
It is virtually insoluble in water (285 mg/l at 30 °C) and insoluble in alcohol. Its Ksp is 1.1 × 10–10. It is soluble in concentrated sulfuric acid. The crystal structure of BaSO4 is known to be rhombic, with a space group pnma.Is barium phosphate soluble in water?
Hydratation of barium and phosphates ions doesn’t yield enough energy to break the bonds in a barium phosphate lattice. So it is ‘insoluble‘. In general salts which high charge ions (+ and -) dissolve slightly in water.
Is bano32 a precipitate?
The barium carbonate will appear in the solid state (as a precipitate) because it is considered insoluble to an aqueous solution. … Therefore, a precipitate is expected to form due to the insolubility of one product which is known as barium carbonate.
Is barium chloride soluble in water?
Barium Chloride is the inorganic compound with the formula BaCl2. It is one of the most common water-soluble salts of barium.
What type of compound is Ba3N2?
Barium nitride (Ba3N2)Is Ba3N2 ionic or covalent?
hence, Ba3N2 is formed with strong ionic bond.
What physical state is barium nitride?Barium nitrate appears as a white crystalline solid. Noncombustible, but accelerates burning of combustible materials.
Article first time published onWhy is barium sulphate not soluble?
The lattice enthalpy of BaO is less than its hydration enthalpy, because of the large size of Ba ions and smaller size of the oxide ion. ions are very large and therefore the lattice enthalpy is much larger than the hydration enthalpy. Therefore BaSO4 is insoluble in water.
Why is barium sulfate not soluble in water?
Whereas, barium sulphate is not at all soluble in water. It is one of the most insoluble ionic salts. This is because the sizes of barium ion and sulphate ion are almost similar and thus the compound has a very high lattice energy which is significantly greater than its hydration energy.
Why is barium oxide soluble but barium sulphate is insoluble in water?
The lattice enthalpy of BaO is less than its hydration enthalpy, because of the large size of Ba ions and smaller size of the oxide ion. Therefore BaO is soluble in water. … Therefore BaSO4 is insoluble in water.
Does barium hydroxide dissociate in water?
Barium hydroxide dissociates completely in water to give barium ions and hydroxyl ions.
Is lithium hydroxide soluble in water?
Lithium hydroxide is an inorganic compound with the formula LiOH(H2O)n. Both the anhydrous and hydrated forms are white hygroscopic solids. They are soluble in water and slightly soluble in ethanol.
Is ag2so4 soluble in water?
Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa). Silver sulfate is the inorganic compound with the formula Ag2SO4. It is a white solid with low solubility in water.
Is bapo4 soluble?
Precipitation of barium phosphate from aqueous solutions of a barium salt and a phosphate salt forms the basis for a number of conclusions drawn in general chemistry. For example, the formation of a solid white precipitate is offered as evidence that barium phosphate is insoluble.
Is fepo4 soluble in water?
Although it is insoluble in water, it is soluble to varying degrees in dilute hydrochloric-acid solutions, such as those in the stomach. The degree of solubility can be controlled by the method of mfr.
Is barium iodide soluble in water?
NamesSolubility in water166.7 g/100 mL (0 °C) 221 g/100 mL (20 °C) 246.6 g/100 mL (70 °C)Solubilitysoluble in ethanol, acetoneMagnetic susceptibility (χ)-124.0·10−6 cm3/molStructure
Is barium chloride soluble in organic solvent?
It is soluble in water, insoluble in acetone, ethanol and ether, slightly soluble in acid, sulfuric acid. Crystallization from aqueous solution of barium chloride often contains two crystal water.
Is barium aqueous?
The chloride, hydroxide, and nitrate of barium are water soluble and are frequently detected in aqueous environments.
Is fe2o3 a covalent compound?
Iron oxide or ferric oxide has three oxygen atoms and two iron atoms. The bond formed between iron and oxygen is due to the difference in electronegativity between the two atoms. … Since iron is metal and oxygen is non-metal therefore the bonding between oxygen and iron is ionic.
How do you write Ba3N2?
Barium nitride (Ba3N2)
Is na2so4 ionic or covalent?
BRIAN M. Sodium Sulfate is an ionic compound formed by two ions, Sodium Na+ and Sulfate SO−24 . In order for these two polyatomic ions to bond the charges must be equal and opposite.
Is barium nitrate soluble in ethanol?
Barium Nitrate for Optical Soluble in water, and insoluble in alcohol.
Is barium carbonate soluble in water?
Barium carbonate is an odorless white inorganic solid. It occurs in nature as the mineral witherite. It is soluble in water at 24 mg/L at 25 °C, soluble in acids (except sulfuric acid) and in ethanol.
How many barium atoms are present in the compound barium nitride?
ElementSymbolNumber of AtomsBariumBa3NitrogeniumN2
Is barium nitrate ionic compound?
Barium nitrate is an ionic compound. Barium is a metal and nitrate is a polyatomic ion, which means it is a group of nonmetals.
Is nh4oh ionic or covalent?
It’s an ionic compound composed of covalently-bound, polyatomic ions.
What is ZnF2?
ZINC fluoride | ZnF2 – PubChem.
What is the name of Li4C?
I read in my chemistry textbook that lithium carbide (Li4C) is the only carbide amongst alkali metals.