How many orbitals can n 3

Mia Kelly
Published Feb 13, 2026
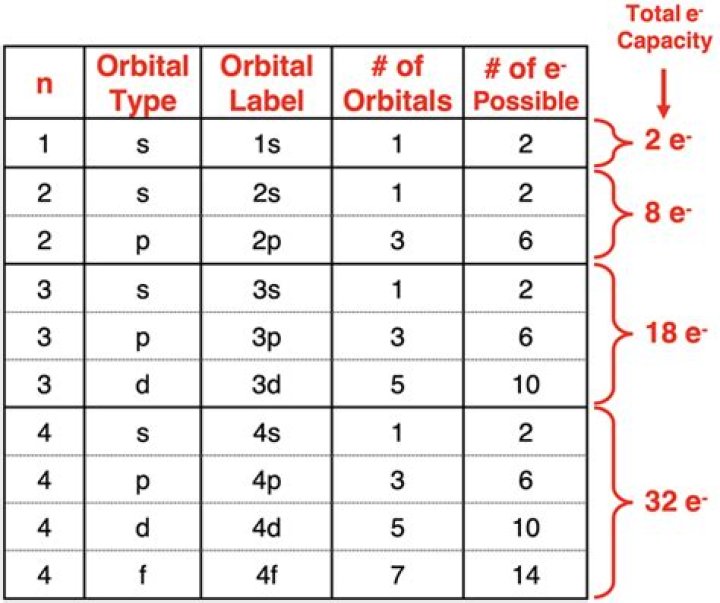
There are nine orbitals in the n = 3 shell. There is one orbital in the 3s subshell and three orbitals in the 3p subshell. The n = 3 shell, however, also includes 3d orbitals.
How many electrons can n 3 hold?
2 electrons are the maximum number of electrons that can exist in any orbital. In the n = 3 shell, 2 electrons max fit into the s subshell and 6 electrons max in the 3px, 3py, and 3pz. The maximum number of electrons that can fit inside a ‘n’ shell is 2n^2. So it is 18.
How many orbitals have n 3 and ML 1 Enter your answer as an integer without units?
Each ml value represents one orbital. So, there are two orbitals that have n = 3 and ml=−1 m l = − 1 .
How many f orbitals can n 3?
As it turns out, none. Each energy level contains a number of subshells given by the angular momentum quantum number, l , which can take values ranging from 0 to n−1 .How many orbitals are present in the Subshells with N 3 L 2?
How many atomic orbitals are there for the subshell with [n = 3, l = 2]? Hint: There are 5 3d orbitals. Discussion: The magnetic q.n. = -2, 1, 0, 1, 2.
How many orbitals are contained in the Third Principal level N 3 of a given atom?
There are 9 orbitals in 3rd energy level.
What Subshells are possible in n 3 energy level?
For n = 3 energy level, the possible values of l are 0, 1 and 2. Thus, there are three subshells namely: l = 0, s subshell ; l = 1, p subshell ; and l = 2, d subshell.
How many electrons in Ni can n 3 and MS?
Since each orbital can hold a maximum of two electrons, the number of electrons that can share the two quantum number n=3 and ml=−2 will be equal to 2 , each having opposite spin.What type of orbitals can have ml =- 3?
s orbitalsf orbitalsl03ml0-3, -2, -1, 0, +1, +2, +3Number of orbitals in designated subshell17
What is the maximum number of electrons that can have n 3 and MS ½?Thus, there are three orbitals associated i.e., 3s,3p and 3d. Now, they are completely filled. Now, it is evident that in any nth orbit, there are a maximum of $2 n^{2}$ electrons. Thus when n=3, maximum number of electrons = 18.
Article first time published onHow many orbitals are there in L 3?
The most complex set of orbitals are the f orbitals. When l = 3, ml values can be −3, −2, −1, 0, +1, +2, +3 for a total of seven different orbital shapes.
How many electrons can fit in the orbital for which N 3 and L 1?
Check Answer and Solution for above questi b) When n= 3, l=0 means 3s orbital which can have 2 electrons . Hence total 6 electrons can fit for n=3 and l=1 If the value of third shell is 3 or n=3 with sub-shell value l=1.
How do you find the number of orbitals N?
To calculate the amount of orbitals from the principal quantum number, use n2. There are n2 orbitals for each energy level. For n = 1, there is 12 or one orbital. For n = 2, there are 22 or four orbitals.
Which energy level is represented by N 3?
Principal Energy Levels and SublevelsPrincipal energy levelNumber of possible sublevelsOrbital Designation by Principal Energy Level and Subleveln = 111sn = 222s 2pn = 333s 3p 3d
What has 3 electrons in energy level n 3?
Orbitals and Electron Capacity of the First Four Principle Energy LevelsPrinciple energy level (n)Type of sublevelMaximum number of electrons (2n2)3s18pd
How many Subshells are in the N 3 shell?
The n = 3 shell, for example, contains three subshells: the 3s, 3p, and 3d orbitals.
When N 3 What is ML and L?
For n = 3, l = 0, 1, 2 For l = 0 ml = 0 For l = 1 ml = -1, 0, or +1 For l = 2 ml = -2, -1, 0, +1, or +2 There are 9 ml values and therefore 9 orbitals with n = 3.
How many quantum states are there in n 3?
distinct orbitals. So, if each electron is described by an unique set of quantum numbers, you can conclude that 18 sets of quantum numbers are possible for the third energy level.
Which is orbital for N 3 and L 2?
This is a 3d orbital, because n =3 and l = 2 which is a d-subshell.
How many sublevels are there with n 3 in an atom?
Explanation: The third electron shell has 3 subshells, which are 3s , 3p , and 3d . An s subshell only has one orbital.
What is the maximum number of electrons that can be associated with the following n 3 L 1'm =- 1?
n=3, l=1, the resultant subshell is 3p. Thus, number of orbitals having n=3, l=1 and m=-1 is one and the maximum number of electrons that can be accommodated in this orbital is two.
What type of orbital is L 3?
That is a d orbital. So for l = 3, the wave function will have three nodes. Those are the f orbitals.
How many orbitals can n 4 have?
l = 3, m1 = –3, –2, –1, 0, 1, 2, 3; seven 4f orbitals. Hence, the number of possible orbitals when n = 4 are sixteen.
How many electrons can have the quantum numbers n 3 and L 2?
– Means the respective orbital where n = 3, l = 2 will be ‘3d’ because 3d orbitals contain five subshells and the principal quantum number is 3. – We know that 3d orbital can accommodate 10 electrons (each subshell can accommodate two electrons). – Therefore the number of electrons are in n = 3, l = 2 are 10.
How many electrons can fit in the orbital for which N 4 and L 3?
1023003 How many electrons can fit in the orbital for which n = 4 and l = 2? CORRECT ANSWER SOLUTION n=3,1 = 1 means that is 3p orbital so it can accommodate maximum 6 electrons.
What is the maximum number of orbitals that can be identified with N 3 L 1 M 0?
Solution: The value of n=3 and l=1 suggests that it is a 3p-orbital while the value f ml=0 [magnetic quantum number ] shows that the given 3p-orbital is 3pz in nature. Hence, the maximum number of orbitals identified by the given quantum number is only 1, i.e. 3pz.
How many orbitals can the following quantum numbers n 3 l 1 ml 0?
A. The value of n=3 and l =1 suggest that it is a 3p orbital while the value of m1 = 0 [magnetic quantum number] shows that the given 3p orbital is 3pz in nature. Hence, the maximum number of orbitals identified by the given quantum number is only 1, i.e. 3pz.
How many number of orbitals are allowed in n shell?
The number of orbitals in ‘N’ shell is 9.
How many electrons can be described by the quantum numbers n 3 and L 1?
So, n=3 and l = 1 indicates electrons are present in the 3p subshell. The p subshell has a maximum of 3 orbitals and each orbital contains a maximum of 2 electrons. Hence, the correct option is (D) 6.
How many electrons are there in the 3rd principal energy level N 3 of a phosphorus atom?
Above that, the third energy level can hold up to 18 electrons.