How many ions are in CaF2

Emma Valentine
Published Feb 13, 2026
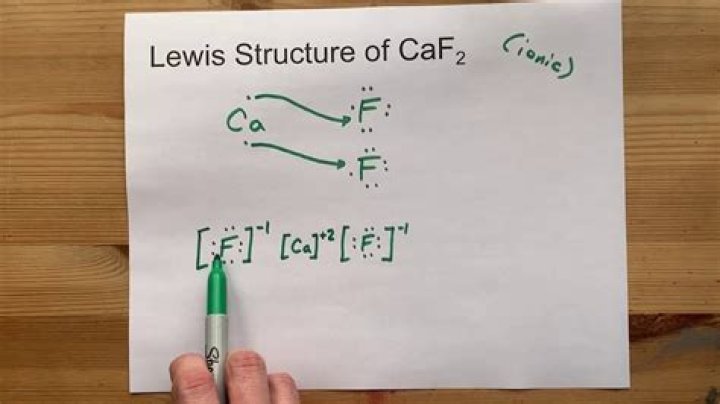
The unit cell has 4 Ca2+ ions and 8 F1- ions.
What is the ionic charge of CaF2?
In calcium fluoride, the calcium ion Ca2+ , has a charge of 2+ , and the fluoride ion F1− has a charge of 1− . We will need two fluoride ions to make the overall charge zero.
What is cn of each ion in CaF2?
In CaF2 the coordination number of Ca2+ is 8. As it is surrounded by 8 F ions. In CaF2 the coordination number of F- is 4. As it is surrounded by 4 Ca ions.
Why CaF2 is ionic?
CaF2 is an ionic compound because, in the CaF2 molecule, calcium acts as a cation by donating its 2 extra electrons from the valence shell, and Fluoride acts as an anion by accepting the electrons from Calcium, completing its octet. Therefore, Calcium and fluoride together form an ionic bond, i.e., CaF2.Is CaF2 ionic?
nglos324 – caf2. Calcium fluoride is an ionic crystal with the fluorine anions in a simple cubic array and calcium cations in half of the cubic sites of the structure.
Is Cl2 ionic or molecular?
In covalent bonds, like chlorine gas (Cl2), both atoms share and hold tightly onto each other’s electrons.
What is the percent composition of CaF2?
For example CaF2 is approximately 50% Calcium ions.
Is CaF2 conductive?
Fluorite is the mineral form of calcium fluoride (CaF2) and is an important host of fluorine. … We found that the electrical conductivity of fluorite is very high at moderate temperature only.Is CaF2 a molecule?
Calcium Fluoride is a polyatomic molecule that contains one calcium molecule and two fluoride molecules. Calcium Fluoride is a quasilinear molecule the bonds are created from the single electrons of calcium and the single electron from fluoride.
Is o2 ionic or covalent?Oxygen forms a covalent bond with itself, since oxygen gas is just two oxygen atoms bonded together with a covalent bond. It also forms covalent bonds…
Article first time published onWhat bond is present in CaF2?
The bonding shows a ‘more ionic’ nature in CaF2. The thermal vibration parameters of the individual atoms of calcium and fluorine have also been studied and reported.
Is N2 ionic or covalent?
N2 has a non-polar covalent bond. As in any homonuclear diatomic, the bonding electrons are shared equally by the two nitrogen atoms.
What is the radius ratio of CaF2?
Calcium fluoride occurs naturally as the mineral fluorite. The ionic radius of the calcium ion is 1.26 Å and that of the fluoride ion is 1.17 Å. … With a radius ratio of 0.929 (essentially unity), the smaller ion is expected to prefer a cubic hole.
What are the coordination number of Ca 2 and F?
In fluorite structure each Ca2+ ion is surrounded by eight F” ions. Thus, the coordination number of Ca2+ is eight.
Is NH3 ionic or covalent?
NH3 has a covalent single bond among its nitrogen and hydrogen atoms. A covalent bond means the N and H atoms share valence electrons while creating…
Is CaF2 an electrolyte or Nonelectrolyte?
Well certainly the calcium fluoride solution is an electrolyte….
Is SiO2 an ionic bond?
And silicon dioxide is most definitely a covalent compound. An ionic compound is any compound that has at least one bond in it that is an ionic bond, that is a bond where the electronegativity difference between the two atoms in the bond is greater than or equal to 1.7.
How do you find the mass of caf2?
The formula mass of CaF 2 is 78.08 u. The formula mass of KNO 3 is 101.10 u.
How do you find the molar mass of caf2?
- Ca = 40.078 grams per mole.
- F = 18.998 grams per mole.
What ion is CL?
Chloride Ion is a chlorine anion that forms the negatively charged part of certain salts, including sodium and hydrogen chloride salts, and is an essential electrolyte located in all body fluids responsible for maintaining acid/base balance, transmitting nerve impulses and regulating fluid in and out of cells.
What kind of bond is Cl and Cl?
The two chlorine atoms are said to be joined by a covalent bond. The reason that the two chlorine atoms stick together is that the shared pair of electrons is attracted to the nucleus of both chlorine atoms.
How many ionic bonds are in Cl2?
The Cl atoms in a Cl2 molecule each attain an octet by sharing their unpaired electrons. There is one bonding pair in Cl2, which lies in the region of overlap of the two octet circles. pairs are called lone pairs. The two atoms have identical electronegativities, so the Cl–Cl bond is a nonpolar covalent bond.
How many ions does fluoride have?
Typically the fluoride anion is surrounded by four or six cations, as is typical for other halides.
What is the conductivity of chlorine?
Thermal ConductivityNameSymbol0.000089 W/cmKChlorineCl0.0000949 W/cmKKryptonKr0.0001772 W/cmKArgonAr0.0002598 W/cmKNitrogenN
What kind of ion is Na+?
Rule 1: Cations. Naming the element and adding the word “ion” forms the cation name. So, Na+ is “sodium ion”.
Is CuCl2 ionic?
The chemical formula shows the compound copper(II) chloride. … Therefore, each copper atom loses two electrons for two chlorine atoms. The result is a copper ion with a charge of positive two, and two chloride ions with a charge of negative one each. Hence, copper(II) chloride is an ionic compound.
Why is o2 an ion?
An atom either loses or gains one or more electrons to form an ion. Each electron carries a charge of −1 whereas each proton within the atomic nucleus carries a charge of +1 . … An electrically-neutral oxygen atom gains two electrons to form an oxygen ion with two negative charges.
What bond exists in li2o and CaF2?
Lithium oxide and calcium fluoride show ionic characters.
How many bonds does chlorine form?
AtomValenceBromine1Chlorine1Iodine1Oxygen2
Is ICl3 ionic or covalent?
ICl3 is made from only nonmetals and is a covalent compound.
Is N2 a compound?
Molecular nitrogen (N2) is a very common chemical compound in which two nitrogen atoms are tightly bound together. Molecular nitrogen is a colorless, odorless, tasteless, and inert gas at normal temperatures and pressures.