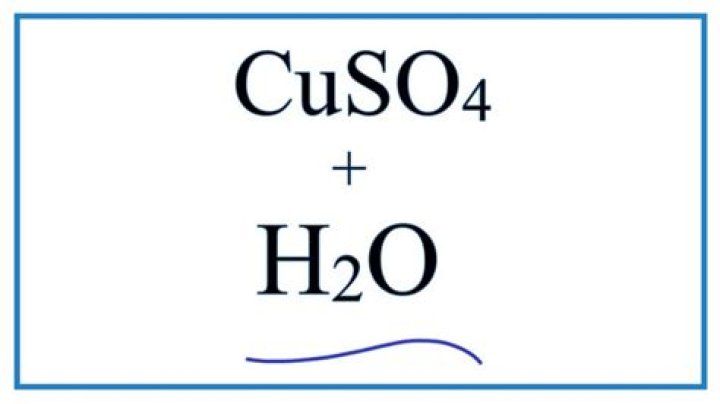
How many h2o are in CuSO4

Emily Dawson
Published Feb 13, 2026
Four water molecules are co-ordinated with Cu2+ and one is hydrogen bonded with SO42- in CuSO4.
What hydrate is CuSO4 5H2O?
Copper sulfate pentahydrate. Copper(II) sulfate pentahydrate is the pentahydrate of copper(2+) sulfate. A bright blue crystalline solid. It is a hydrate and a metal sulfate.
How do you calculate the percent composition of water?
Divide the mass of the water lost by the mass of hydrate and multiply by 100. The theoretical (actual) percent hydration (percent water) can be calculated from the formula of the hydrate by dividing the mass of water in one mole of the hydrate by the molar mass of the hydrate and multiplying by 100.
What percentage of water h2o is found in the hydrate na2s ● 9h2o?
Sodium sulfate decahydrate is 55.9%H2O .What is %H2O in CuSO4 5H2O?
molecular weight of the compound. H2O % in CuSO4.5H2O = 5 x 18 x 100 / 250. = 36 % Therefore, hydrated copper sulphate contains 36% water molecules. Hope it helps!
What is the color of CuSO4 * 5H2O?
CuSO4· 5H2O is blue in colour whereas anhydrous CuSO4 is colourless.
How many hydrogen bonded H2O are there in CuSO4 5H2O?
9 How many hydrogen bonded water molecules are associated in CuSO4. 5H2O ? Solution Only one water molecule, which is outside the coordination sphere, is hydrogen bonded.
What is the percentage composition of na2s 23.5 h2o?
ElementSymbolMass PercentSodiumNa27.349%HydrogenH5.995%OxygenO47.583%SulfurS19.073%How do you calculate percent by mass?
To calculate the mass percent of an element in a compound, we divide the mass of the element in 1 mole of the compound by the compound’s molar mass and multiply the result by 100.
What is the percent by mass of water in the hydrate na2so4 10h2o GFM of the anhydrous na2so4 is 142 g mol?The percent by mass of water in Na2 SO4 x 10H2 O is 55.91%.
Article first time published onWhat is the percent water in the hydrated salt niso4 ⋅ 7h2o report to 4 sig fig?
The percent of water is 44.91 % (m/m).
What is the percent composition of C6H12O6?
Percent Composition An elemental analysis of a white crystalline compound believed to be C6H12O6, analyzed for C, H and O gave the following results. C = 40.00 % , H = 6.71 %, O = 53.28 % by wt.
How do you find the percent of H in H2O?
To find the mass percent of hydrogen in water, take the molar mass of hydrogen in the water molecule, divide by the total molar mass of water, and multiply by 100. Dividing 2.016 by 18.016 gives you 0.1119. Multiply 0.1119 by 100 to get the answer: 11.19 percent.
How do you calculate percentage of water crystallization?
Once we have the relative mass formula of a hydrated compound, we can determine how much of this mass is water of crystallisation. Multiply the degree of hydration by the M r of water, and divide this by the M r of the whole compound.
What type of reaction is CuSO4 5H2O?
ReactionBalanced Chemical ReactionType of Chemical ReactionCuSO4(s) + H2O(l) → CuSO4*5H2O(s)CuSO4(s) + 5H2O(l) → CuSO4*5H2O(s)SynthesisCa(s) + S(s) → CaS(s)Ca(s) + S(s) → CaS(s)SynthesisMg(s) + O2(g) → MgO(s)2Mg(s) + O2(g) → 2MgO(s)Synthesis
How much water is in a mol?
This is stated: the molar mass of water is 18.02 g/mol. Notice that the molar mass and the formula mass are numerically the same. Objective 2: Use molar mass to convert between mass in grams and amount in moles of a chemical compound.
What is the difference between CuSO4 and CuSO4 5H2O?
The key difference between CuSO4 and CuSO4 5H2O is that CuSO4 is amorphous, whereas CuSO4 5H2O is crystalline. CuSO4 is the chemical formula of copper(II) sulfate, while CuSO4 5H2O is the hydrated form of copper(II) sulfate.
Is there hydrogen bond in CuSO4 5H2O?
CuSO4. 5H2O has one hydrogen – bonded molecule of water.
How many water molecules are involved in hydrogen bonding in bacl2 2h2o?
2 water molecules are attached here.
How many water molecules are associated with crystalline oxalic acid?
Second, the DQCC ( χ CLS ) and asymmetry parameter ( η CLS ) have been calculated for a cluster extracted from the solids and composed of an oxalic acid molecule surrounded by four close neighboring water molecules (see Fig. 3).
What is the colour of 5H2O?
5H2O is blue . The colour of any vol. of water or 5H2O is colourless .
What is the colour of CuSO4 without water?
In hydrated CuSO4 the water molecules surrounding the Central metal (Cu) function as ligands which bring d-d transition and hence emits blue colour in visible region due which hydrated CuSO4 appears blue and as anhydrous CuSO4 doesn’t have any water of crystallisation hence remains white in colour.
Why CuSO4 5H2O is blue in colour while CuSO4 is Colourless?
In CuSO4. 5H2O water acts as ligand as a result it causes crystal field splitting. Hence d—d transition is possible in CuSO4. … In the anhydrous CuSO4 due to the absence of water ligand crystal field splitting is not possible and hence no colour.
How can I calculate percentage?
Percentage can be calculated by dividing the value by the total value, and then multiplying the result by 100. The formula used to calculate percentage is: (value/total value)×100%.
What is percentage by volume?
Volume/Volume Percentage Definition Volume/volume percentage (v/v percent) is a measure of the concentration of a substance in a solution. It is expressed as the ratio of the volume of the solute to the total volume of the solution multiplied by 100.
How do you find the percent by volume?
Volume Percent: The volume percent is used to express the concentration of a solution when the volume of a solute and the volume of a solution is given: Volume Percent=Volume of SoluteVolume of Solution×100%
What happens when anhydrous copper sulphate is added to water?
Water being added to some anhydrous copper (II) sulphate. The anhydrous form is pale green in colour. When water is added slowly, the copper sulphate becomes hydrated, with five water molecules binding to each copper sulphate molecule. This pentahydrate form is blue.
What happens when anhydrous copper sulphate is sprinkled with water?
Anhydrous copper sulphate gets hydrated and turns blue on addition of a few drops of water.
What is the percentage of calcium in calcium carbonate?
Therefore, the percentage of calcium in calcium carbonate is 40.04%.
What is the percentage composition of Na2S2O3?
ElementSymbolMass PercentSodiumNa29.081%OxygenO30.358%SulfurS40.561%
What is the symbol for sodium?
sodium (Na), chemical element of the alkali metal group (Group 1 [Ia]) of the periodic table. Sodium is a very soft silvery-white metal. Sodium is the most common alkali metal and the sixth most abundant element on Earth, comprising 2.8 percent of Earth’s crust.