How hard is stoichiometry

Rachel Hickman
Published Feb 12, 2026
Stoichiometry can be difficult because it builds upon a number of individual skills. To be successful you must master the skills and learn how to plan your problem solving strategy. Master each of these skills before moving on: Calculating Molar Mass.
Is stoichiometry the hardest part of chemistry?
Stoichiometry is arguably one of the most difficult concepts for students to grasp in a general chemistry class. Stoichiometry requires students to synthesize their knowledge of moles, balanced equations and proportional reasoning to describe a process that is too small to see.
What type of math is stoichiometry?
“The Math of Chemical Reactions—Stoichiometry” Tutorial Summary : Stoichiometry uses the principles of dimensional analysis to use information about one species in a chemical reaction to determine information about a different species in the same reaction.
Why does a math student find stoichiometry hard to solve?
From the foregoing discussion, it has been shown that students have difficulties in stoichiometry problem-solving as a result of lack of understanding of a number of concepts related to stoichiometry that influence their ability to solve stoichiometry problems.Which Chemistry topic is the hardest?
Most of the time the difficult topics in chemistry are General organic chemistry, Ionic and Chemical equilibrium, thermodynamics, nuclear physics and electrochemistry.
How do we use stoichiometry in real life?
Stoichiometry is at the heart of the production of many things you use in your daily life. Soap, tires, fertilizer, gasoline, deodorant, and chocolate bars are just a few commodities you use that are chemically engineered, or produced through chemical reactions.
How do you master stoichiometry?
- Balance the equation.
- Convert units of a given substance to moles.
- Using the mole ratio, calculate the moles of substance yielded by the reaction.
- Convert moles of wanted substance to desired units.
What is the point of stoichiometry?
Stoichiometry measures these quantitative relationships, and is used to determine the amount of products and reactants that are produced or needed in a given reaction. Describing the quantitative relationships among substances as they participate in chemical reactions is known as reaction stoichiometry.What is the purpose of stoichiometry how is it important to chemists and chemistry?
Why Stoichiometry Is Important You can’t understand chemistry without grasping the basics of stoichiometry because it helps you predict how much of a reactant participates in a chemical reaction, how much product you’ll get, and how much reactant might be left over.
What is studied in stoichiometry?Stoichiometry is the study of the relative quantities of reactants and products in chemical reactions and how to calculate those quantities.
Article first time published onWhat is stoichiometry quizlet?
Stoichiometry. (chemistry) the relation between the quantities of substances that take part in a reaction or form a compound (typically a ratio of whole integers) Limiting Reactant. the reactant that limits the amounts of the other reactants that can combine and the amount of product that can form in a chemical …
Is chemistry or biology harder?
As a general rule, most students find biology easier except, they may be required to memorize more information. Chemistry is usually more difficult, especially the labs, because they require a better understanding of mathematics, especially error analysis.
Is physics harder than chemistry?
Physics is slightly harder than chemistry because it is more math-oriented and has more abstract concepts. … During high school, most students find chemistry more difficult than physics. 20% more students get a 3 or higher in their AP physics exams as compared to their chemistry exams.
Is a chemistry major harder than biology?
The easy answer is yes, chemistry is harder than biology. I don’t have a quantifiable reason, but from my experience in college and from teaching chemistry and biology in high school, students find the math needed for chemistry to be challenging and it requires higher levels of critical reasoning.
Is BCA good for Career?
Career Prospects BCA graduates have good job prospects both in the government and private sector companies. After successfully passing their BCA course, students can easily find lucrative job opportunities in leading IT companies like Oracle, IBM, Infosys and Wipro.
Which subjects in BCA course?
- Fundamentals of Computers.
- C Programming.
- Operating Systems.
- Multimedia Systems.
- Understanding Organisational Behaviour.
- Data and Database Management Systems.
- Web-Based Application Development.
- Computer Lab and Practical Work.
What is BCA and its scope?
A BCA graduate has a great scope in jobs as a Web Designer, System Manager, Software Developer, Computer Programmer, Web Developer, Software Developer, software tester, etc. depending on the skills acquired by the student at the time of BCA.
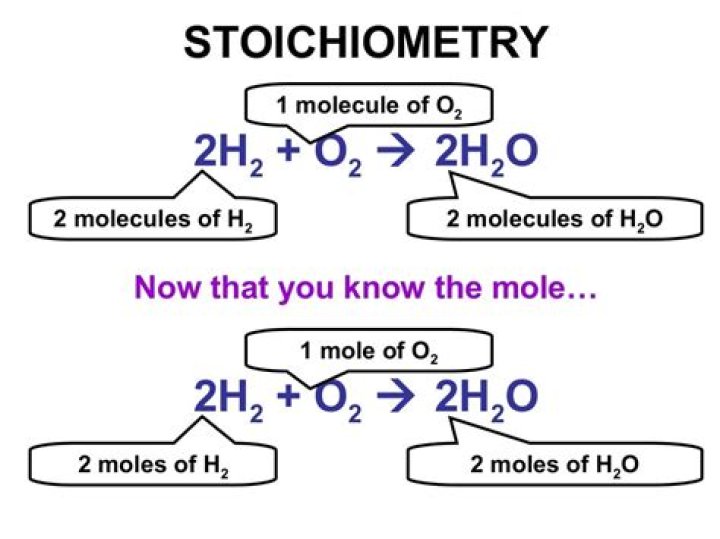
What is a mole in stoichiometry?
The mole is a key unit in chemistry. The molar mass of a substance, in grams, is numerically equal to one atom’s or molecule’s mass in atomic mass units. … A balanced chemical reaction gives equivalences in moles that allow stoichiometry calculations to be performed.
How many moles are in Fe2O3?
The molar mass of Fe2O3 is 159.7 g/mol.
What is the mole ratio?
Mole Ratio: is a conversion factor between compounds in a chemical reaction, that is derived from the coefficients of the compounds in a balanced equation. The mole ratio is therefore used to convert between quantities of compounds in a chemical reaction.
How many hydrogens are needed for 10 moles of ammonia?
Thus 15 moles of hydrogen are required to produce 10 moles of ammonia.
What careers use stoichiometry?
- Baking.
- Pharmaceutical industry.
- Hair Dressers.
- Mining companies.
- Engineering.
How is stoichiometry used in food industry?
If you want to get the right amount of the product, you need to measure the specific amounts of each reactant (ingredient) as given in the recipe, such as flour and sugar. … Stoichiometry tells us how much of each reactant is required to obtain the desired amount of product.
Why is stoichiometry so important for chemical engineering?
Stoichiometry is important for chemical engineering because it tells engineers how materials interact, react, and play off of one another.
How can stoichiometry be used in the future?
Stoichiometry predicts the amount of product produced For example, we used stoichiometry to determine how many s’more “reactants” we would need to make 10 s’mores. We can also use stoichiometry to predict how much product we’ll get with the amount of each reactant we have.
Why would a nurse use stoichiometric calculations?
Nurses routinely use stoichiometry to convert the drip ratios of medication to the prescribed dosages. They use a persons weight and conversion factors to determine the correct dosage of medicine to give. If the stoichiometry calculations are done incorrectly the patient can potentially be harmed.
Do coefficients matter in stoichiometry?
Stoichiometric coefficients must be added to make the equation balanced. In this example, there are only one sulfur atom present on the reactant side, so a coefficient of 2 should be added in front of H2SO4 to have an equal number of sulfur on both sides of the equation.
How does the stoichiometry of solutions affect our daily living?
Stoichiometry continues to be useful in many walks of life—a farmer determining how much fertilizer to use, figuring out how fast you have to go to get somewhere in a specific period of time, or just to make conversions between systems like Celsius and Fahrenheit.
What is stoichiometric combustion?
We call a stoichiometric combustion a combustion without excess or lack of air, where all available oxygen is completely consumed. Assuming that nitrogen reacts only in negligible proportions and is found after. combustion in molecular form, the general equation of a stoichiometric combustion.
Who discovered stoichiometry?
Reaction Stochiometry Movie Text. Stoichiometry was first discovered by Jeremias Richter, a German chemist. It was Richter who coined the term stoichiometry, a tongue-twisting word that baffles students to this day. Stoichiometry was derived from stoikheion, Greek for “element”, and “metron”, meaning measure.
What are stoichiometric defects?
Stoichiometric defects are those defects in which the ratio of cations to anions remains the same are represented by the molecular formula.