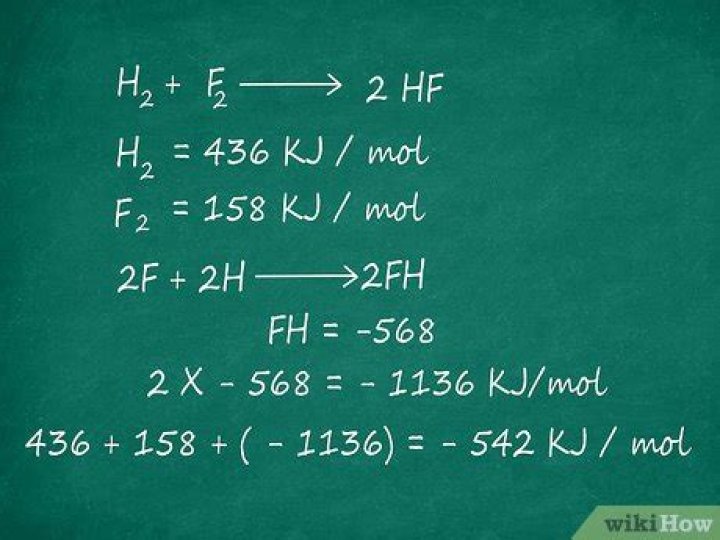
How enthalpy is calculated

Mia Morrison
Published Apr 09, 2026
Use the formula ∆H = m x s x ∆T to solve. Once you have m, the mass of your reactants, s, the specific heat of your product, and ∆T, the temperature change from your reaction, you are prepared to find the enthalpy of reaction. Simply plug your values into the formula ∆H = m x s x ∆T and multiply to solve.
Is the dissolution of urea endothermic or exothermic?
The process is endothermic. The decrease in temperature indicates that the process for the dissolution of urea in water requires energy.
What is molar enthalpy?
Molar enthalpy of fusion is the amount of energy needed to change one mole of a substance from the solid phase to the liquid phase at constant temperature and pressure. It is also known as the molar heat of fusion or latent heat of fusion. Molar enthalpy of fusion is expressed in units of kilojoules per mole (kJ/mol).
Is entropy or enthalpy the driving force for the dissolution of urea?
Dissolving cellobiose in water and the urea solution absorb heat, which is an entropy-driven process. Dissolving cellobiose in NaOH solution and mixed NaOH/urea solution is exothermic, which is an enthalpy-driven process. OH− plays an important role in the dissolving process by forming a hydrogen-bonding complex.How do you calculate the enthalpy of a solution?
- Amount of energy released or absorbed is calculated. q = m × Cg × ΔT. q = amount of energy released or absorbed. …
- calculate moles of solute. n = m ÷ M. …
- Amount of energy (heat) released or absorbed per mole of solute is calculated. ΔHsoln = q ÷ n.
What happens if urea dissolve in water?
Urea remains urea, it only dissociates in the presence of some enzymes. When urea is mixed with water this creates a cold (endothermic) reaction. … On addition to the soil, urea dissolves into the soil solution and supplies ammonium form of N, but it is rapidly converted to nitrate in the soil.
What is the enthalpy of dissolution of urea?
5. The standard integral enthalpy of solution of urea in water determined by us is 15.39 kJ mol-I whereas the data of Malec and Sarnowski (9) 15.41 kJ mol-I; togwinienko (lo), 15.49 kJ mol-I; de Visser et al.
Is dissolution of urea in water adsorption?
Urea is a molecular compound, so it does not dissociate into ions. The dissolution process involves energy to break the attractive bonds and forces between the molecules in the compound and releasing energy as the molecules form new attractive forces with the water molecules.Is enthalpy or entropy the driving force?
The change in enthalpy and change in entropy of a reaction are the driving forces behind all chemical reactions. In this lesson, we will examine a new function called free energy, which combines enthalpy and entropy and can be used to determine whether or not a given reaction will occur spontaneously.
Is entropy and enthalpy the same?Difference Between Enthalpy and EntropyEnthalpy is a kind of energyEntropy is a propertyIt is the sum of internal energy and flows energyIt is the measurement of the randomness of moleculesIt is denoted by symbol HIt is denoted by symbol S
Article first time published onWhat is the molar enthalpy of combustion of pentane?
Phase behaviorGas propertiesStd enthalpy change of formation, ΔfHogas–146.8 kJ/molStandard molar entropy, Sogas347.82 J/(mol K)Enthalpy of combustion, ΔcHogas–3535 kJ/mol
What is enthalpy in chemistry?
enthalpy, the sum of the internal energy and the product of the pressure and volume of a thermodynamic system. … According to the law of energy conservation, the change in internal energy is equal to the heat transferred to, less the work done by, the system.
How do you calculate the enthalpy of enthalpy of formation?
This equation essentially states that the standard enthalpy change of formation is equal to the sum of the standard enthalpies of formation of the products minus the sum of the standard enthalpies of formation of the reactants. and the standard enthalpy of formation values: ΔH fo[A] = 433 KJ/mol.
What do you mean by enthalpy of solution?
The enthalpy of solution, enthalpy of dissolution, or heat of solution is the enthalpy change associated with the dissolution of a substance in a solvent at constant pressure resulting in infinite dilution.
What is the difference between Q and Delta H?
Q is the energy transfer due to thermal reactions such as heating water, cooking, etc. anywhere where there is a heat transfer. You can say that Q (Heat) is energy in transit. Enthalpy (Delta H), on the other hand, is the state of the system, the total heat content.
Why is urea in water endothermic?
– Urea dissolves in water very easily. It is very easily soluble in water because of the hydrogen bonds, as a result of its formation with water. … Urea dissolution is an endothermic process. So, on addition to water liquid becomes cold thus halting the dissolution process.
Why is the dissolution of urea in water endothermic?
Urea dissolution into water as an endothermic reaction is simply the thermodynamic result of the net energy (in this case a positive enthalphy of reaction) of breaking apart the crystalline matrix and the resulting energy of the solvation of the molecule in water.
Does urea sublime?
Urea crystallizes in long needles or prisms which melt at 132° C. and sublime when heated in vacuo. … When heated above its melting-point, it yields ammonia, cyanuric acid, biuret and ammelide.
Is urea polar or nonpolar?
Urea has two C-N single bonds and one C=O. double bond at an angle of about 120 degrees. This gives rise to a dipole moment which makes it a polar molecule.
Is urea an oxidizing agent?
Special Hazards of Combustion Products: Irritating ammonia gas may be formed in fire. UREA HYDROGEN PEROXIDE is an oxidizing agent. Liable to spontaneous combustion when heated or in contact with organic materials.
Why can't urea get wet?
Urea is lost for the following reason. It reacts with water to form ammonium carbonate. … However, if the urea is on the soil surface when the breakdown occurs, the ammonia gas is lost to the atmosphere. These losses can be greater if the soil has a high pH or if the soil is wet when the urea is applied.
Is urea soluble in alcohol?
It is noticeable that the solubility of urea in a mixture of ethanol and water increases with increasing temperature and decreases with increasing ethanol concentration to the mixture. The fact that the solubility decreases with decreasing temperature at low pressures is attributed to the reduction in density 19.
How is urea converted to nitrogen?
When urea is applied, an enzyme in soil and plant residue called urease quickly converts the urea into ammonia N. If this conversion occurs beneath the soil surface, the ammonia is converted to ammonium nitrogen and bound to soil particles. That’s a good thing.
How do you know if something is enthalpy or entropy driven?
If a reaction’s ΔH is positive, and ΔS is negative, the reaction is always thermodynamically disfavored. … Reactions can be ‘driven by enthalpy‘ (where a very exothermic reaction (negative ΔH) overcomes a decrease in entropy) or ‘driven by entropy’ where an endothermic reaction occurs because of a highly positive ΔS.
What is enthalpy and entropy?
Enthalpy is the amount of internal energy contained in a compound whereas entropy is the amount of intrinsic disorder within the compound.
Does burning fuel increase entropy?
The entropy of the system increases during a combustion reaction. The combination of energy decrease and entropy increase dictates that combustion reactions are spontaneous reactions.
Is urea soluble in HCl?
Urea is soluble in H2O, ethanol, methanol, glycerol and Conc. HCl.
How much urea can you dissolve in water?
Urea will dissolve in its own weight of water, but becomes more difficult to dissolve as the concentration increases. Urea is endothermic. As urea dissolves, it causes the temperature of the solution falls, so a practical limit of 25 – 30% w/v (25 to 30 kg of urea in 100 litres of solution) is suggested.
What kind of molecule is urea?
Urea is a nitrogenous compound containing a carbonyl group attached to two amine groups with osmotic diuretic activity. In vivo, urea is formed in the liver via the urea cycle from ammonia and is the final end product of protein metabolism.
How do you find enthalpy from entropy?
Entropy ( S ) defines the degree of randomness or disorder in a system. where at constant temperature, the change on free energy is defined as: ΔG=ΔH−TΔS . Therefore, the free energy expression provides a relationship between enthalpy and entropy. Thus, for a system at equilibrium, ΔG=0 , and then we find that ΔS=ΔHT .
Does enthalpy increase with entropy?
The reason for this relationship is that if energy is added to or released from the system, it has to be partitioned into new states. Thus, an enthalpy change can also have an effect on entropy. … In an exothermic reaction, the external entropy (entropy of the surroundings) increases.