How does sulfur affect pH

Rachel Hickman
Published Feb 12, 2026
The cheapest way to lower the soil pH is to add elemental sulfur to the soil. Soil bacteria change the sulfur to sulfuric acid, lowering the soil pH. If the soil pH is greater than 5.5, apply elemental sulfur (S) to decrease the soil pH to 4.5 (see Table 1).
How much does sulfur lower pH?
An unfounded soil management suggestion is that soil pH can be successfully reduced by applying moderate rates of elemental sulfur (about 100 to 200 lb/acre elemental S).
Does sulfate increase pH?
Sulfate added directly to the soil does not alter soil pH because soil is acidified by hydrogen ions that are released during the chemical reaction that transforms sulfur into sulfate.
Is sulfur acidic or alkaline?
Sulfur is an alkaline that helps in neutralizing the body acid. This is because it enhances metabolic processes.Can sulfur lower soil pH?
Elemental sulfur can be applied as a soil amendment to decrease the pH or acidify such soils.
How much sulfur is needed to lower pH in lawn?
Some distributors and soil testing labs will recommend applying sulfur to reduce the soil pH of turf when it is over 7.5. Unfortunately, you can only apply 5 lbs sulfur/1000 sq. ft. per application to avoid burning turfgrass mowed at 2 inches or more, and a total of 10 lbs sulfur/1000 sq.
What is the pH of Sulphur dioxide?
The rate of solution of the gas was rapid for the first V2 hr during which time the pH decreased from 6.1 to approximately 4.0. As the rate of solution of atmospheric sulfur dioxide decreased the pH of the water decreased to a final value of 3.8.
Is Sulphur an acid?
SulfurAtomic propertiesOxidation states−2, −1, 0, +1, +2, +3, +4, +5, +6 (a strongly acidic oxide)ElectronegativityPauling scale: 2.58How long does it take Sulphur to lower pH?
The soil acidification process depends on microorganisms’ activity and soil aeration, thus the reaction is slow in cold, wet soils. In normal conditions, it is expected that pH will decrease within three to six months after the application of elemental sulfur or ammonium-based fertilizers.
What is difference between Sulphur and sulfur?Sulphur is the preferred spelling British English. In summary, sulfur is the American spelling, and sulphur the British spelling of this word.
Article first time published onHow does sulfur change soil pH?
The cheapest way to lower the soil pH is to add elemental sulfur to the soil. Soil bacteria change the sulfur to sulfuric acid, lowering the soil pH. If the soil pH is greater than 5.5, apply elemental sulfur (S) to decrease the soil pH to 4.5 (see Table 1).
What is difference between sulphur and sulphate?
Sulphur is a chemical element that has only sulphur and does not have a charge. … Sulphate is made from sulphur and oxygen. It is an anion with the chemical formula SO42-. It is the salt of sulphuric acid.
Does sulfur make soil acidic?
You can use elemental sulfur (sold as soil sulfur at most nurseries) to make soil more acidic (lower pH). It’s best if the sulfur can be mixed in the soil before planting. … In moist, warm soil, it will take several months for soil bacteria to convert the sulfur to sulfuric acid.
Is too much sulfur bad for plants?
Some sulfur is beneficial for plants, but if used in excess the sulfur will form excessive salts that can easily kill the plants that you are trying to help. … The main concern when the pH is high is that some plant essential nutrients will not be available for uptake by the root system.
How do you acidify soil pH?
Simply add an inch or two (2.5-5 cm.) of peat to the topsoil in and around plants, or during planting. For another quick fix, water plants several times with a solution of 2 tablespoons vinegar to a gallon of water. This is a great way to adjust pH in container plants.
What causes high pH in soil?
Vegetation type impacts soil pH. … These changes are caused by a loss of organic matter, removal of soil minerals when crops are harvested, erosion of the surface layer, and effects of nitrogen and sulfur fertilizers. Addition of nitrogen and sulfur fertilizers can lower soil pH over time.
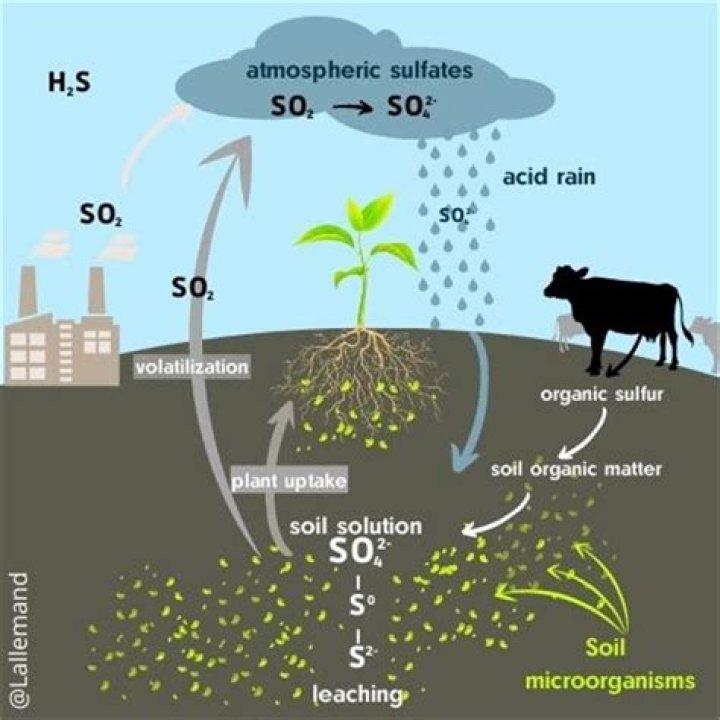
How does sulfur dioxide affect the pH of rain?
Acid rain results when sulfur dioxide (SO2) and nitrogen oxides (NOX) are emitted into the atmosphere and transported by wind and air currents. The SO2 and NOX react with water, oxygen and other chemicals to form sulfuric and nitric acids. These then mix with water and other materials before falling to the ground.
What happens when Sulphur reacts oxygen?
Sulfur reacts with oxygen to form sulfur dioxide. When nonmetals react with one another, the product is a molecular compound. … Sulfur can also combine with oxygen to produce sulfur trioxide.
Is Sulphur trioxide acidic or basic?
SO3 (Sulfur trioxide) is a Lewis acid mainly because the sulfur atom in SO3 has only three electron regions. The sulfur atom tends to accept an electron pair. Besides, SO3 is also a non-metal oxide which is acidic in nature.
What is the fastest way to lower pH in soil?
Soil pH can be reduced most effectively by adding elemental sulfur, aluminum sulfate or sulfuric acid. The choice of which material to use depends on how fast you hope the pH will change and the type/size of plant experiencing the deficiency.
How do I fix high pH in my lawn?
If the pH is too high, your grass cannot properly absorb nutrients. Ideal pH should be between 6.5-7.0, slightly acidic. Generally, lime is added to raise the pH and sulfur is added to lower the pH, and adding compost can naturally correct your pH.
What fertilizer will lower pH of soil?
As previously mentioned, if a lower pH is required, ammonium or urea-based fertilizers can aide in the reduction of pH.
Does Epsom salt lower pH?
Although soil amendment with elemental sulfur lowers soil pH levels through the release of hydrogen ions into the soil, Epsom salt does not release hydrogen ions, so it has no effect on pH.
Does lime increase pH?
Adding lime (Figure 1) increases soil pH (reduces acidity), adds calcium (Ca) and/or magnesium (Mg), and reduces the solubility of Al and Mn in the soil.
What happens when soil pH is too high?
Soil pH is vital to plant health. … A pH reading that is too high or low will lead to a loss of these microorganisms, which will result in a less healthy soil overall. In addition, pH affects the solubility and potency of certain toxic chemicals, such as aluminum, which can be taken up by plants if the pH is off.
What is the purpose of sulfur?
Your body needs sulfur to build and fix your DNA and protect your cells from damage that can lead to serious diseases such as cancers. Sulfur also assists your body to metabolize food and contributes to the health of your skin, tendons, and ligaments. The two amino acids that include sulfur are methionine and cysteine.
What are 3 interesting facts about sulfur?
- Sulfur is an essential element.
- You can find it in amino acids and proteins, which are in the food we eat.
- The element sulfur is also known as brimstone.
- Sulfur’s atomic number is 16.
- There is no sulfur chemical formula because it is not a metal. …
- Sulfurs symbol in the periodic table is S.
Is sulfur highly reactive?
sulfur (S), also spelled sulphur, nonmetallic chemical element belonging to the oxygen group (Group 16 [VIa] of the periodic table), one of the most reactive of the elements. It reacts with all metals except gold and platinum, forming sulfides; it also forms compounds with several nonmetallic elements. …
Is sulfur with F or PH?
Nature Chemical Biology may say that an author’s favorite color is gray, but Nature Physics would say that an author’s favourite colour is grey. In a similar vein, the UK journals — including Nature itself — spell sulphur with ‘ph’ and the US titles spell sulfur with an ‘f’.
What is the difference between sulfur and sulfuric acid?
The key difference between sulfuric acid and sulfurous acid is in the oxidation number of Sulfur. Moreover, when we compare two acids in terms of acidity, sulfuric acid is more acidic than sulfurous acid.
Which is right sulphur or sulfur?
Spelling of the name of the element labelled S. Sulphur or Sulfur? … British English spelt it “sulphur” while North American English used the phonetic spelling “sulfur”. In the late 20thC, the International Union of Pure and Applied Chemistry (IUPAC) decided that the single spelling of sulfur was prefered.



