How does Hunds rule work

Mia Kelly
Published Feb 24, 2026
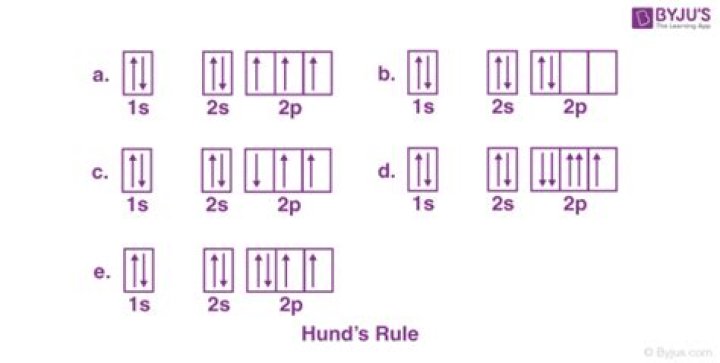
Hund’s rule states that orbitals of equal energy are each occupied by one electron before any orbital is occupied by a second electron and that each of the single electrons must have the same spin. The Figure below shows how a set of three p orbitals is filled with one, two, three, and four electrons.
Why is Hund's rule obeyed?
Hund’s rule states that electron orbitals that are degenerate (same energy) are first half-filled before they are totally-filled. When we are distributing electrons among the molecular orbitals, we do it as shown below: Hence, Hund’s rule is obeyed.
Which element does not follow Hund's rule?
Electrons first fill the degenerate orbitals of similar energy. The first electron filled can be either spin up or spin down. Chromium and copper elements violate Hund’s rule.
Why do we have to spread out electrons using Hund's rule?
Hund’s Rule states that every orbital in a sublevel is singly occupied before any orbital is doubly occupied and all of the electrons in singly occupied orbitals have the same spin. Electrons arrange themselves in order to minimize their interaction energy.What is a violation of Hund's rule?
Hund’s rule states that each subshell in an orbital must be filled with one electron each before anyone is doubly occupied and the spin of all electrons in singly occupied shells is the same. … This type of electronic configuration violated the Hund’s rule.
Why Hund's rule of maximum multiplicity is so called?
The rule states that for a given electron configuration, the lowest energy term is the one with the greatest value of spin multiplicity. This implies that if two or more orbitals of equal energy are available, electrons will occupy them singly before filling them in pairs.
What is the orbital notation for radon?
The ground state electron configuration of ground state gaseous neutral radon is [Xe]. 4f14.
What is the difference between Hund's rule and Pauli exclusion principle?
Hund’s Rule states that if 2 or more degenerate (i.e. same energy) orbitals are available, one electron goes into each until all of them are half full before pairing up . … The Pauli Exclusion Principle states that no two electrons can be identified by the same set of quantum numbers.Is Hunds rule obeyed in MO theory?
Hund’s rule is obeyed. electrons are assigned to orbitals of successively higher energy. … the combination of two atomic orbitals creates only one molecular orbital.
When an electron gets closer to the nucleus?As an electron gets closer to the nucleus the (attraction/repulsion) to the nucleus gets (strongery weaker). 16. For an electron to move from an energy level close to the nucleus to an energy level far from the nucleus it would need to gain lose) energy.
Article first time published onWhy is it important when writing electron configurations?
Knowledge of the electron configuration of different atoms is useful in understanding the structure of the periodic table of elements. This is also useful for describing the chemical bonds that hold atoms together. In bulk materials, this same idea helps explain the peculiar properties of lasers and semiconductors.
What is the highest energy level?
Energy LevelSublevelsMaximum Number of Electrons per Energy Level1s22s8p3s18
Which is correct statement in case of Hund's rule?
Answer: According to Hund’s rule, during filling of electron in the orbitals, every orbital will be singly occupied first before pairing takes place. Therefore maximum number of unpaired electron will be (2l+1) for a given subshell.
What is the Pauli violation?
The Pauli Exclusion Principle states that, in an atom or molecule, no two electrons can have the same four electronic quantum numbers. … Otherwise they will have the same four quantum numbers, in violation of the Pauli Exclusion Principle.
How many orbitals are in the N 3?
There are nine orbitals in the n = 3 shell. There is one orbital in the 3s subshell and three orbitals in the 3p subshell. The n = 3 shell, however, also includes 3d orbitals. The five different orientations of orbitals in the 3d subshell are shown in the figure below.
Which of the following is correct on the basis of Hund's rule but not follow Pauli's principle?
According to Pauli’s exclusion principle, no two electrons have the same set of quantum numbers. Means, if there are two electrons in the same subshell; same orbital then their spin will be different. … Clearly, Hund’s rule and Pauli’s exclusion principle is being followed. So, option IV is also correct.
How many 6p electrons are in RN?
The atomic number of radon is 76 , which means that a neutral atom would also contain 76 electrons. The noble gas shorthand electron configuration is [Xe]4f145d106s26p6 .
What group is ar?
Group18−189.34°C, −308.81°F, 83.81 KPeriod3−185.848°C, −302.526°F, 87.302 KBlockp0.001633Atomic number1839.95State at 20°CGas40Ar
Is lead an atom or molecule?
Lead is a chemical element – a substance that contains only one type of atom. Its official chemical symbol is Pb, and its atomic number is 82, which means that a lead atom has 82 protons in its nucleus.
Is Hund's rule applicable in excited state?
Excited states Hund’s rules work best for the determination of the ground state of an atom or molecule. They are also fairly reliable (with occasional failures) for the determination of the lowest state of a given excited electronic configuration.
What is Hund's rule of maximum multiplicity explain by taking example of carbon?
Hund’s rule for maximum multiplicity: The Hund rule defines that for a given E.C, the term of lowest energy is the one with the highest value of spin multiplicity. This suggests that if two or extra orbitals of the same energy are obtainable, electrons will occupy them singly before satisfying them in pairs.
What is the meaning of maximum multiplicity?
During the filling of orbitals the maximum number of unpaired electrons is achieved and hence the maximum total spin state. This is the maximum multiplicity.
Which has highest bond length?
The carbon–carbon (C–C) bond length in diamond is 154 pm. It is generally considered the average length for a carbon–carbon single bond, but is also the largest bond length that exists for ordinary carbon covalent bonds.
Does the Pauli exclusion principle apply to MOs explain?
The Pauli exclusion principle states that the maximum number of electrons occupying an orbital is two, with opposite spins. Hund’s rule states that when there are several MOs with equal energy, the electrons occupy the MOs one at a time before two occupy the same MO.
What is Lcao in chemistry?
A linear combination of atomic orbitals or LCAO is a quantum superposition of atomic orbitals and a technique for calculating molecular orbitals in quantum chemistry.
What is Pauli's exclusion principle explain the importance of this principle for the understanding of atomic structure?
Pauli’s exclusion principle states that no two electrons in an atom can have all the same quantum numbers. The structure of the periodic table of elements can be explained in terms of the total energy, orbital angular momentum, and spin of electrons in an atom.
Do electrons and protons repel each other?
Project the animation Protons and Electrons. Explain to students that two protons repel each other and that two electrons repel each other. But a proton and an electron attract each other. … Since opposite charges attract each other, the negatively charged electrons are attracted to the positively charged protons.
What color has the highest energy?
Complete answer: In our case of visible light, the highest frequency color, which is violet, which means it will have the highest energy.
Do electrons attract to the nucleus?
The electrons are attracted to the nucleus by the electrostatic force of attraction to the protons. Surprisingly enough, although the minimum energy classically would have the electrons all located at the nucleus (point of strongest attraction) they aren’t all there.
Which electron configuration represents a violation of Hund's rule?
You have two electrons in one 2p orbital, but none in the other 2p orbitals. This violates Hund’s Rule: There must be one electron with the same spin in each orbital of the same energy before you can put two in the same orbital. The electrons in the half-filled 4d orbitals don’t all have the same spin.
What is a sublevel?
Definition of sublevel : a level that is lower than or subordinate to another level a sublevel garage The 60 words were divided into nine separate groups based on grade levels and sublevels.—



