How does Grahams law work

Emma Valentine
Published Apr 01, 2026
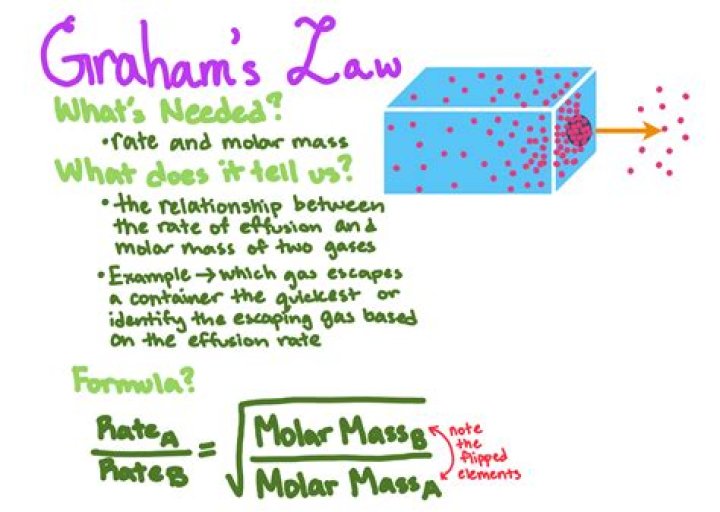
Graham’s law states that the rate of diffusion or of effusion of a gas is inversely proportional to the square root of its molecular weight. … Therefore, the rates of diffusion of different gases are inversely proportional to the square roots of their mass densities.
What is a real life example of Graham's law?
Balloons The escaping of gas molecules through the small opening in the balloon represents the effusion process. The time taken by the gas molecules to completely escape and the balloon to get properly deflated can be calculated with the help of Graham’s law of effusion or diffusion.
Do lighter gases effuse faster?
Explanation: The rate of effusion of a gas is inversely proportional to the square root of the molecular weight of the gas. The lighter a gas is, the faster it will effuse; the heavier a gas is, the slower it will effuse.
How do you use Graham's law of effusion?
From Graham’s law, we can use the molar mass of each gas: rate of effusion of hydrogenrate of effusion of oxygen=√32g mol−1 √2g mol−1 =√16√1=41 rate of effusion of hydrogen rate of effusion of oxygen = 32 g mol − 1 2 g mol − 1 = 16 1 = 4 1 Hydrogen effuses four times as rapidly as oxygen.Which gas is more ideal at STP?
The real gas that acts most like an ideal gas is helium. This is because helium, unlike most gases, exists as a single atom, which makes the van der Waals dispersion forces as low as possible. Another factor is that helium, like other noble gases, has a completely filled outer electron shell.
What is Graham's Law & application?
Grahams law of diffusion : At the given temperature and pressure the rate of diffusion of a gas is inversely proportional to square root of its density or molar mass or vapour density. Applications :→ This principle is used to detect unknown molar mass of a gas.
Why is Grahams law useful?
Graham’s law provides a basis for separating isotopes by diffusion—a method that came to play a crucial role in the development of the atomic bomb. Graham’s law is most accurate for molecular effusion which involves the movement of one gas at a time through a hole.
How much faster does hydrogen gas effuse than neon gas?
This ratio is designed to compare rates. So hydrogen gas effuses 3.16 times faster than neon.What does Avogadro's principle state?
Avogadro’s law, a statement that under the same conditions of temperature and pressure, equal volumes of different gases contain an equal number of molecules.
How do you calculate the ratio of effusion?Background: The ratio of the rates of effusion of two gases is equal to the square root of the inverse ratio of their molar masses or densities. The effusion rate of a gas is inversely proportional to the square root of its molar mass.
Article first time published onHow does temperature affect Graham's law?
Diffusion is faster at higher temperatures because the gas molecules have greater kinetic energy. … Graham’s Law states that the effusion rate of a gas is inversely proportional to the square root of the mass of its particles.
Does pressure affect speed of gas particle?
The Kinetic-Molecular Theory Explains the Behavior of Gases, Part I. … If the volume is held constant, the increased speed of the gas molecules results in more frequent and more forceful collisions with the walls of the container, therefore increasing the pressure (Figure 1). Charles’s law.
Do gas particles move slow?
In gases the particles move rapidly in all directions, frequently colliding with each other and the side of the container. … The actual average speed of the particles depends on their mass as well as the temperature – heavier particles move more slowly than lighter ones at the same temperature.
Why does C2H6 deviate from ideal behavior?
The C2H6(g) deviates much more from ideal behavior than the CH4(g)does. Which of the following best helps explain this deviation? C2H6 molecules have a larger, more polarizable electron cloud than CH4 molecules do.
Why is neon an ideal gas?
Gases are most ideal at high temperature and low pressure. The ideality of a gas also depends on the strength and type of attractive forces that exist between the particles. neon is more ideal than water vapor because neon’s atoms are only attracted by weak dispersion forces.
Why is hydrogen an ideal gas?
Hydrogen and helium are the closest to ideal gases because they have both the least amount of excluded volume (thereby bringing its molar volume close to that of an ideal gas), and the weakest intermolecular attractions.
What is constant in Charles Law?
Charles’s Law states that the Volume (V) of a gas is directly proportional to the temperature (T). This law is valid as long as the pressure and the amount of gas are constant. The temperature must be an absolute temperature: VT=k(constant) The constant, k, will depend on the number of moles and the pressure.
What are the examples of effusion?
Effusion is defined as a loss of material across a boundary. A common example of effusion is the loss of gas inside of a balloon over time. The rate at which gases will effuse from a balloon is affected by a number of factors.
Is effusion and diffusion the same?
Diffusion occurs when gas molecules disperse throughout a container. Effusion occurs when a gas passes through an opening that is smaller than the mean free path of the particles, that is, the average distance traveled between collisions.
How much faster does helium escape argon?
Helium escapes faster than Ar. It does so at 3.16 times the rate of the argon.
Which is wrong according to kinetic theory?
There are no attractive forces between the molecules of gas.
How can molar mass be calculated?
To calculate the molar mass of a compound, calculate the molar mass of each element in the compound, then multiply the element’s atomic mass by the number of atoms of that element in the compound. Add the molar masses of each element in the compound to calculate the molar mass of the compound!
What is a real life example of Avogadro's law?
Example of Avogadro’s Law in Everyday Life The best example of Avogadro’s law is blowing up a balloon. The balloon’s volume increases as you add moles of gas. Similarly, when you deflate a balloon, gas leaves the balloon and its volume shrinks.
How do you use Avogadro's law?
Avogadro’s Law is in evidence whenever you blow up a balloon. The volume of the balloon increases as you add moles of gas to the balloon by blowing it up. If the container holding the gas is rigid rather than flexible, pressure can be substituted for volume in Avogadro’s Law.
What is Graham's Law of diffusion Class 11?
Graham’s law is that the rate of diffusion of the gas is inversely proportional to the square root of the molecular mass of that gas. Rate of diffusion of the gas ∝ 1√M1. M is the molecular mass of the gas.
How do you calculate the molar mass of an unknown gas?
First the ideal gas law will be used to solve for the moles of unknown gas (n). Then the mass of the gas divided by the moles will give the molar mass. Step 2: Solve. Now divide g by mol to get the molar mass.
Which gas diffuses faster oxygen or nitrogen?
nitrogen because it has low relative molecular mass than oxygen. so because of low molecular mass it is lighter and diffuses faster.
Why nitrogen gas diffuses faster at higher temperature?
Answer: Since an increase in temperature represents an increase in the average molecular speed, diffusion occurs faster at higher temperatures.
Which gas Effuses faster hydrogen or chlorine How much faster?
Hydrogen effuses approximately 6 times as fast as chlorine.
What ratios are compared in Graham's law?
Graham’s law of diffusion states that the ratio of the diffusion rate of two gases is the same as the ratio of the square root of the molar mass of the gases.
What does rate of effusion depend on?
The effusion rate for a gas depends directly on the average velocity of its particles. Thus, the faster the gas particles are moving, the more likely they are to pass through the effusion orifice.