How does CI become an ion

Sophia Edwards
Published Mar 01, 2026
In chemical ionization (CI) ions are formed by reaction of the sample molecules with a known preselected set of reagent ions. These reagent ions are produced by ion–molecule reactions in a reagent gas introduced in the ion source of the mass spectrometer at a pressure of 0.1 to 1.0 Torr.
What does CI mean in molecules?
Chemical Ionization (CI) is a “soft” ionization technique that produces ions with little excess energy. As a result, less fragmentation is observed in the mass spectrum. Since this increases the abundance of the molecular ion, the technique is complimentary to 70 eV EI.
How isobutane and methane ionized the sample?
The reagent gas (methane, isobutane, or ammonia) is present in the ion source at a pressure of 1 torr. It is ionized by an electron beam and the resulting ions undergo a complex series of ion-molecule reactions to produce species such as CH5+ in methane.
How do the spectra for EI and CI differ from one another?
How do the spectra differ for EI, field ionization, and CI sources differ from one another? The most fragementation and thus the most complex spectra are encountered with electron impact ionization. … Chemical and electron impact ionization result in higher sensitivities than does field ionization.What is CI in mass spectroscopy?
Chemical ionization (CI) is a soft ionization technique used in mass spectrometry. … Reagent gas molecules are ionized by electron ionization, which subsequently react with analyte molecules in the gas phase in order to achieve ionization.
What is CI in the periodic table?
Chlorine – Element information, properties and uses | Periodic Table.
What ion is chloride?
Chloride Ion is a chlorine anion that forms the negatively charged part of certain salts, including sodium and hydrogen chloride salts, and is an essential electrolyte located in all body fluids responsible for maintaining acid/base balance, transmitting nerve impulses and regulating fluid in and out of cells.
What is CL in Chem?
chlorine (Cl), chemical element, the second lightest member of the halogen elements, or Group 17 (Group VIIa) of the periodic table. Chlorine is a toxic, corrosive, greenish yellow gas that is irritating to the eyes and to the respiratory system.What does CI mean in biology?
Confidence interval (CI), usually 95% CI. Inferential. A range of values you can be 95% confident contains the true mean.
What ions does EI form?EI produces a mixture of positive and negative ions, as well as neutral species. Positive-ion EI mass spectra are more commonly recorded because these ions form more readily.
Article first time published onWhat is ionization electron?
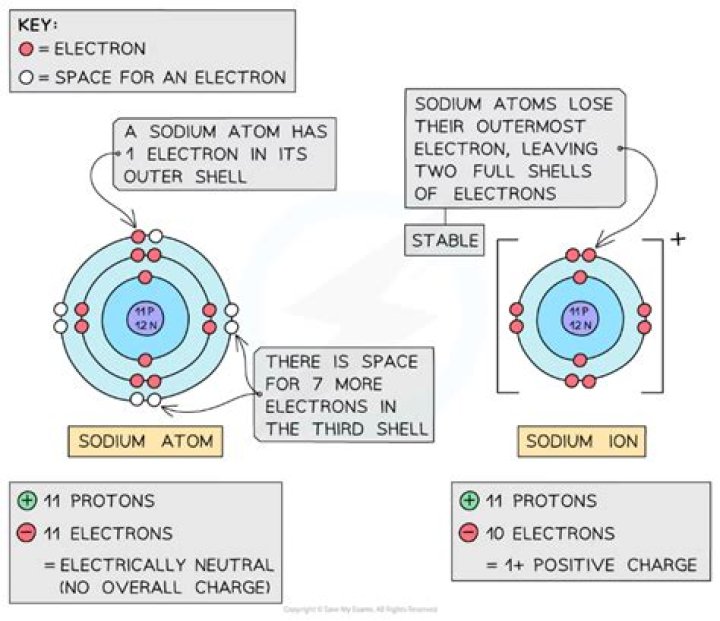
Ionization or ionisation is the process by which an atom or a molecule acquires a negative or positive charge by gaining or losing electrons, often in conjunction with other chemical changes. … The resulting electrically charged atom or molecule is called an ion.
Why is 70 eV used in electron ionization?
If enough energy is transferred by the process, the molecule will eject a valence electron and form a radical cation (M+). Since the ionization is produced by a single electron that is accelerated to 70 V, this is commonly referred to as 70 eV EI.
What is field ionization?
Field ionization (FI) is the ionization of a gaseous molecule by an intense electric field, usually created by a sharp electrode at a high potential.39.
What is Methylpropane used for?
2-methylpropane is used in the production of alkylate petrol and isobutylene as well as in the manufacture of pharmaceutical and cosmetic products. Due to the ozone-diminishing effects of freons, this substance is gradually replacing them in aerosols and as coolants in refrigerators.
How does Fast Atom Bombardment work?
Fast atom bombardment (FAB) is an ionization technique used in mass spectrometry in which a beam of high energy atoms strikes a surface to create ions. … When a beam of high energy ions is used instead of atoms (as in secondary ion mass spectrometry), the method is known as liquid secondary ion mass spectrometry (LSIMS).
Which is the molecular peak in mass spectroscopy?
IsotopeZMass16O1615.9949
Is chloride a CL or CI?
The chloride ion /ˈklɔːraɪd/ is the anion (negatively charged ion) Cl−. It is formed when the element chlorine (a halogen) gains an electron or when a compound such as hydrogen chloride is dissolved in water or other polar solvents.
What is the name of Ca2+?
Ca2+ is just calcium ion, not calcium(II) ion, because calcium only has one kind of stable cation.
What is the charge of C?
NumberElementCharge6carbon4+7nitrogen3-8oxygen2-9fluorine1-
Is Cl A metal?
Chlorine is a non-metal. … Chlorine usually accepts electrons so is an oxidant, thus it shows the property of a non-metal. The element chlorine is a yellowish-green gas which is extremely reactive and react with various metals.
How many electrons does CL have?
The chlorine atom will also have 17 electrons, as an atom is neutral.
Why is chlorine CL and not ch?
Answer: Chlorine was discovered by Humphry Davy in the early 19th century and was named after the Greek word ‘chloros’ which meant that it had to be differentiated from carbon with the addition of either h or l after the case of carbon.
What does CI mean in medical?
Abbrev for CONFIDENCE INTERVAL.
What is the value of CL?
The first scale of electronegativity was developed by Linus Pauling and on his scale chlorine has a value of 3.16 on a scale running from from about 0.7 (an estimate for francium) to 2.20 (for hydrogen) to 3.98 (fluorine).
Can you calculate the electron ionization?
Decide how many electrons the atom contains. This number is the same as Z unless the atom has already lost some electrons. Calculate the ionization energy, in units of electron volts, for a one-electron atom by squaring Z and then multiplying that result by 13.6.
What is the ionization energy of CL?
Ionization EnergyName chemical elementSymbol12,9676ChlorineCl13,5984HydrogenH13,6181OxygenO13,9996KryptonKr
What is ionization in chemistry class 10?
In chemistry, ionization is a process where an atom or a molecule, that is electrically neutral, is converted into electrically charged atoms by gaining or losing an electron from the outer shell. The resulting atom is called the ion.
When substance is bombarded with electrons of energy 9 to 15 eV by the loss of one electron ion is produced?
(1) MOLECULAR ION PEAK: When a sample substance is bombarded with electrons of 9 to 15 eV energies, the molecular ion (M+) is produced by loss of a single electron. M+ e ‘ M+ +2e-, where M represents molecule & M+ is the molecular ion /parent ion.
How does a mass spectrometer work?
A mass spectrometer can measure the mass of a molecule only after it converts the molecule to a gas-phase ion. To do so, it imparts an electrical charge to molecules and converts the resultant flux of electrically charged ions into a proportional electrical current that a data system then reads.
What is desorption in mass spectrometry?
Laser desorption mass spectrometry uses a laser to ionize the analyte. In some cases, the laser can be applied directly to the sample. It will remove molecules from the surface of the material and then ionize them. … This technique is called matrix-assisted laser desorption ionization or MALDI.
What does Maldi Tof measure?
During MALDI-TOF analysis, the m/z ratio of an ion is measured by determining the time required for it to travel the length of the flight tube. A few TOF analyzers incorporate an ion mirror at the rear end of the flight tube, which serves to reflect back ions through the flight tube to a detector.