How does a fractionating tower work

Emily Dawson
Published Apr 14, 2026
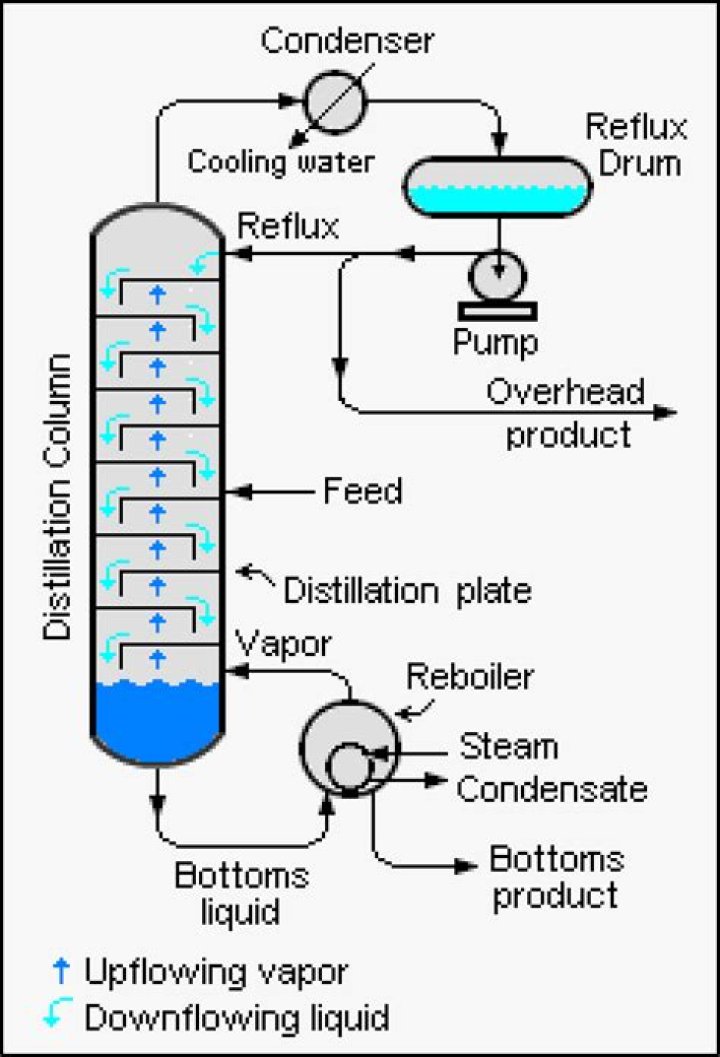
In a fractional distillation, a mixture of liquids is boiled and the resulting vapors travel up a glass tube called a “fractionating column” and separate. The fractionating column is placed between the flask containing the mixture and the “Y” adaptor and improves the separation between the liquids being distilled.
How does a fractionation tower work?
Fractionating columns help to separate the mixture by allowing the mixed vapors to cool, condense, and vaporize again in accordance with Raoult’s law. … The vapor condenses on glass spurs (known as trays or plates) inside the column, and returns to the distilling flask, refluxing the rising distillate vapor.
How does fractional distillation work step by step?
- heated crude oil enters a tall fractionating column , which is hot at the bottom and gets cooler towards the top.
- vapours from the oil rise through the column.
- vapours condense when they become cool enough.
- liquids are led out of the column at different heights.
How does a distillation tower work?
The distillation column is made up of a series of stacked plates. A liquid feed containing the mixture of two or more liquids enters the column at one or more points. The liquid flows over the plates, and vapor bubbles up through the liquid via holes in the plates.What are the 5 steps of fractional distillation?
- Evaporation. Crude oil is heated until it evaporates. Crude oil vapour is put into a fractionating column at the bottom and rises upwards.
- Condensation. The temperature is highest at the bottom of the column. …
- Collection. The fractions are collected.
What is the function of distillation?
Distillation is used to separate liquids from nonvolatile solids, as in the separation of alcoholic liquors from fermented materials, or in the separation of two or more liquids having different boiling points, as in the separation of gasoline, kerosene, and lubricating oil from crude oil.
What is a fractional distillation tower?
Fractional distillation is the process by which oil refineries separate crude oil into different, more useful hydrocarbon products based on their relative molecular weights in a distillation tower. …
How does fractional distillation separate compounds?
Fractional distillation is the separation of a mixture into its component parts, or fractions. Chemical compounds are separated by heating them to a temperature at which one or more fractions of the mixture will vaporize.How do packed columns work?
Packed columns are used for distillation, gas absorption and liquid-liquid extraction. The gas-liquid contact in a packed column is continuous, not stage-wise, as in a plate column. The liquid flows down in the column over a packing surface and the vapor (or the gas) moves counter-currently, up the column.
How does fractional distillation separate hydrocarbons into fractions?Crude oil is separated by fractional distillation. Crude oil is heated to vaporize the different hydrocarbons in a tank which is cool at the top and hot at the bottom. The vapours then rise and the different hydrocarbons condense at their specific boiling points, allowing them to be separated.
Article first time published onWhat is distillation flask?
The distilling flask, also known as a fractional distillation flask or fractioning flask, is a vessel with a round bottom and a long neck from which a side arm protrudes. … The placement of the side arm along the neck varies depending on the characteristics of the solution to be distilled.
Which liquid will get distilled first?
On account of least molecular mas , liquid H2 has the weakest intermolecular interaction. Thus , it has minimum boiling point and is distilled first.
What is the function of fractionating column Class 9?
The fractionating column allows the vapours of the mixture to cool, condense and evaporate again by Raoult’s law. The fractionating column allows the continuous condensation and vaporization of the mixture and enriches the separation of a component.
Which fraction leaves at the top of the tower?
Answer: correct answer is option number a) Refinery gases…
Why is fractional distillation done?
Fractional distillation is a process by which components in a chemical mixture are separated into different parts (called fractions) according to their different boiling points. Fractional distillation is used to purify chemicals and to separate mixtures to obtain their components.
Why is fractional distillation important?
Fractional distillation is useful for separating a mixture of substances with narrow differences in boiling points, and is the most important step in the refining process. … For example, only 40% of distilled crude oil is gasoline; however, gasoline is one of the major products made by oil companies.
How is fractional distillation different from simple distillation?
The main difference between fractional distillation and simple distillation is that simple distillation separate liquids with boiling point gaps of at least 50 degrees whereas fractional distillation separates liquids with closer boiling points.
How does a simple distillation work?
Simple distillation works because the dissolved solute has a much higher boiling point than the solvent. When the solution is heated, solvent vapour evaporates from the solution. The gas moves away and is cooled and condensed .
Is distilled water to drink?
Is Distilled Water Safe to Drink? Distilled water is safe to drink. But you’ll probably find it flat or bland. That’s because it’s stripped of important minerals like calcium, sodium, and magnesium that give tap water its familiar flavor.
How do we use distillation in real life?
Distillation is the process of separating components of a mixture based on different boiling points. Examples of uses of distillation include purification of alcohol, desalination, crude oil refining, and making liquefied gases from air.
What is tower packing?
Tower packing is a mass of inert, solid shapes. Within a cylindrical column, it provides a greater surface area for the gas and liquid in a web scrubber to make contact.
How does random packing work?
Random packing uses a random distribution of small packing materials to assist in the separation process, while structured packing uses larger, fixed packing structures. These more formal materials guide the liquid materials through complex structural channels into a specific, fixed shape.
What are packed towers?
The packed tower is a continuous contact type gas-liquid mass transfer equipment, which makes the two-phase composition change continuously along the tower height. In the normal condition, the gas phase presents continuous phase while the liquid phase presents dispersed phase.
How many stages does a distillation column have?
When more ideal stages are required, the packing should be separated into sections of no more than 10 ideal stages, with vapor and liquid redistribution in between.
How do you clean a distillation column?
To clean the inside of a still, fill the boiler with a gallon of white vinegar, attach the column, and boil for about an hour. After boiling the vinegar for an hour, carefully dump out the the vinegar. It will be HOT- we recommend using heat resistant gloves.
Why are distillation columns important?
Distillation columns are used to separate mixed feed streams into their own distinct products. The last 50 years have shown tremendous improvement in our understanding of industrial distillation equipment and systems. Advanced technology is taking over in the design, control, and operation of towers.
How does fractional distillation separate ethanol and water?
For example, liquid ethanol can be separated from a mixture of ethanol and water by fractional distillation. This method works because the liquids in the mixture have different boiling points. When the mixture is heated, one liquid evaporates before the other.
How does chromatography work?
Chromatography is a method of separating mixtures by using a moving solvent on filter paper. … The solvent flows along the paper through the spots and on, carrying the substances from the spot. Each of these will, if the solvent mixture has been well chosen, move at a different rate from the others.
Can you separate alcohol and water by freezing?
Once fermentation is complete, a wine’s alcohol content remains stable throughout its life, unless you do something drastic, like boil it, or, in a more complicated system, freeze it. It’s easier to extract water from wine by freezing it than to extract alcohol from wine by freezing it.
What is fractional distillation Class 9?
In simple words, fractional distillation is the process of separation of a mixture into fractions by their boiling points by heating them to a temperature at which one or more fractions vaporize. Thus, fractional distillation is based on the property that the boiling point of each fraction is different.
What is a distillation setup?
The distillation setup consists of a flask (solution), a three-way adapter, a water-jacketed condenser, a vacuum adapter and a round-bottom flask to catch the liquid that condenses. … The setup has to have a pressure release opening (usually the vacuum adapter). Otherwise teh entire setup will blow apart upon heating.