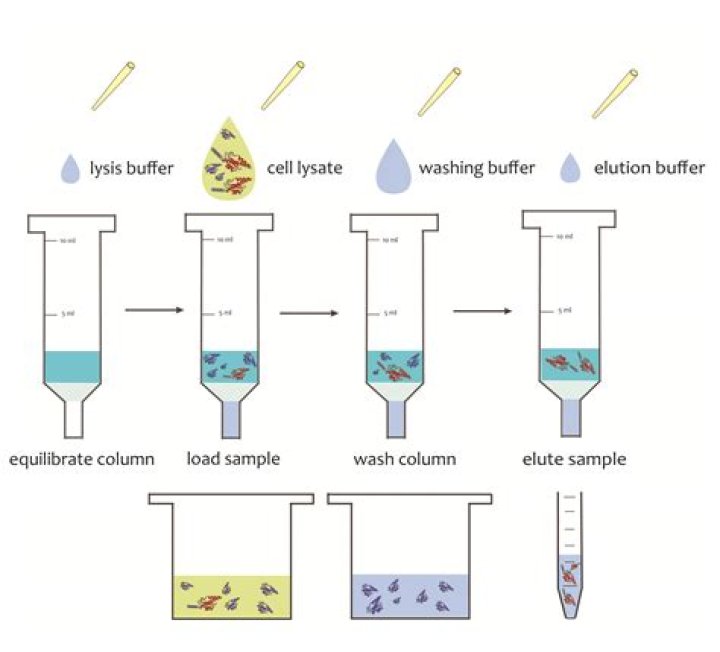
How do you make a lysis buffer

Mia Morrison
Published Apr 13, 2026
BufferBuffer Range (pH)Phosphate buffer5.8 – 8
What are the ingredients of lysis solution?
The major components of the lysis buffer for blood DNA extraction are Tris, EDTA, MgCl2, KCl, NaCl and SDS.
How do you make a RIPA lysis buffer?
Preparation of RIPA lysis buffer: • 10mM Tris-HCl, pH 8.0 • 1mM EDTA • 0.5mM EGTA • 1% Triton X-100 • 0.1% Sodium Deoxycholate • 0.1% SDS • 140mM NaCl • Dilute with dH2O • This solution is stable at room temperature. Add 1mM PMSF immediately before use.
What are the 2 components of the lysis solution?
Cell lysis buffer for RNA extraction is highly denaturing and is usually composed of phenol and guanidine isothiocyanate. RNase inhibitors are usually present in the lysis buffer, since RNases can be very resistant to denaturation and remain active. For extraction of DNA the lysis buffer will commonly contain SDS.How do you make a buffer for DNA extraction?
Teacher Preparation: Prepare the DNA extraction buffer. In a container, add 900mL of water, 50mL of dishwashing detergent (or 100mL shampoo), and finally 2 teaspoons of salt. Slowly invert the bottle to mix the extraction buffer. Note: A modification can be made based on the needs of the students.
How do you make Tris EDTA buffer?
- Measure out 1 mL 1M Tris-Cl (pH 8.0) and add to a 100 mL Duran bottle.
- Measure out 0.2 mL 0.5M EDTA (pH 8.0) and add to the Duran bottle.
- Top up the solution to 100 mL by adding 98.8 mL of distilled water.
- Place the lid on the bottle and invert a few times to mix.
What is lysis buffer What is the composition of lysis buffer?
Most lysis buffers contain buffering salts (e.g. Tris-HCl) and ionic salts (e.g. NaCl) to regulate the pH and osmolarity of the lysate. Sometimes detergents (such as Triton X-100 or SDS) are added to break up membrane structures.
What is SDS in lysis buffer?
SDS also denatures most of the proteins in the cells, which helps with the separation of the proteins from the plasmid later in the process. It is important during this step to make sure that the re-suspension and lysis buffers are well mixed, although not too vigorously (see below).How do you prepare a NP 40 lysis buffer?
The Unfolded Protein Response and Cellular Stress, Part A Prepare 1% NP40 lysis buffer (LB). (To make 50 ml of 1% NP40 lysis buffer, add 5 ml of 10% NP40, 500 μl of 1 mM EDTA, 1.5 ml of 150 mM NaCl, and 1 ml of 20 mM Tris–Cl, pH 7.4; and bring the total volume to 50 ml using dH2O.)
Why is EDTA in lysis buffer?Lysis buffer contains ethylenediaminetetraacetic acid (EDTA) as EDTA is a metal chelator. … The EDTA has a higher affinity for chelating Mg2+ ions compared to EGTA, therefore in many situations, EDTA is preferred.
Article first time published onHow do you make a 1x lysis buffer?
- Measure out 3 mL sodium chloride (5 M), 5 mL Tris-HCl (1 M, pH 8.0), 1 mL nonidet P-40, 5 mL sodium deoxycholate (10 %), 1 mL SDS (10%) and add to a 100 mL Duran bottle.
- Top up the Duran bottle to 100 mL with ddH2O.
How do you lyse mammalian cells?
The freeze-thaw method is commonly used to lyse bacterial and mammalian cells. The technique involves freezing a cell suspension in a dry ice/ethanol bath or freezer and then thawing the material at room temperature or 37°C.
How do you lyse cells using lysis buffer?
Aspirate off liquid. Gently resuspend the cell pellet in ice cold cell lysis buffer (with fresh protease inhibitors), use 1 ml buffer for 107 cells. Incubate cells for 30 minutes on ice. If needed, sonicate the lysates on ice for 15-30 seconds to disrupt genomic DNA and cellular components.
How is lysis buffer prepared for protein extraction?
- Prepare a 100 mM solution in double distilled water.
- Set pH to 9.0 with HCl.
- Boil until colorless. …
- Cool to room temperature.
- Set pH to 9.0 again.
- Boil until colorless.
- Repeat this cycle until the solution remains at pH 9.0 after boiling and cooling.
Is CTAB a lysis buffer?
Cationic detergent cetyltrimethylammonium bromide (CTAB) is used to liberate and complex with total cellular nucleic acids. This buffer contains 2% w/v CTAB, 20 mM EDTA.Na₂.
What are the 4 steps of DNA extraction?
To extract DNA the four steps in order are lysis, separation, precipitation, and purification. The lysis step opens up cells that contain DNA. After…
Why is imidazole in lysis buffer?
Ni-NTA Lysis Buffer contains 10 mM imidazole to minimize binding of untagged, histidine rich contaminating proteins and increase purity with fewer wash steps. If the tagged protein does not bind under these conditions, the amount of imidazole should be reduced to 1-5 mM.
How do you make Tris HCl buffer?
- Solution A: Dissolve 121.14 g Tris (American Bioanalytical #AB14042) in 800 ml dH2O.
- Adjust pH to 7.0 with the appropriate volume of concentrated HCl. Bring final volume to 1 liter with deionized water.
- Autoclave and store at room temperature.
How do you make a 50 millimeter Tris base?
- Buffer and Media. Elution buffer 50 mM Tris-HCl pH 7.5.
- Materials. •
- Dissolve Tris base in 1 L of double distilled water.
- Add NaCl and imidazole to the solution.
- Adjust pH to 7.5 with HCl solution.
How do you make citrate buffer?
- Prepare 800 mL of distilled water in a suitable container.
- Add 24.269 g of Sodium Citrate dihydrate to the solution.
- Add 3.358 g of Citric Acid to the solution.
- Adjust solution to desired pH using 0.1N HCl (typically pH ≈ 6.0).
How do you make Tris NaCl buffer?
Tris-buffered saline (TBS) is an excellent wash buffer for many types of immunoassays. To make 1 L of 10X TBS stock solution, dissolve 24 g Tris and 88 g NaCl in 900 mL of water and then adjust the pH to 7.6 and final volume to 1 L.
How does Tris buffer work?
Tris is the main buffering component; its chief role is to maintain the pH of the buffer at a stable point, usually 8.0. Additionally, tris likely interacts with the LPS (lipopolysaccharide) in the membrane, serving to destabilize the membrane further.
How do you dilute NP-40?
Directions: 1) Mix 20 ml of NP-40 with 80 ml of ddH2O by stirring. 2) Store at 4°C. Note: Nonidet P-40 is chemically identical to Igepal-CA-630.
What is NP-40 lysis buffer?
NP40 Cell Lysis Buffer is a high-quality, ready-to-use lysis buffer suitable for the preparation of cell extracts for ELISA, western blotting, and antibody bead immunoassays (Luminex) applications. … The formulation helps retain the protein structure and function needed for enzyme assays or immunoassays.
What is the function of NP-40 detergent?
NP-40 is often used to break open all membranes within a cell, including the nuclear membrane. To break only the cytoplasmic membrane, other detergents such as IGEPAL CA-630 can be used. NP-40 has applications in paper and textile processing, in paints and coatings, and in agrochemical manufacturing.
How do you make an alkaline lysis solution 1?
Prepare Solution I from standard stocks in batches of approx. 100 ml, autoclave for 15 minutes at 15 psi and store at 4°C. 1. 10 N NaOH stock solution (50 mL) Dissolve 20 gram of NaOH in 50 mL sterilized de-ion water.
Why is SDS used in lysis buffer?
Using 1% SDS in lysis buffer ensures complete solubilization and denatures phosphatases.
Why is SDS used in lysis?
Strong ionic detergents such as sodium dodecyl sulphate (SDS) are able to provide cell lysis of the order of seconds, tending to denature proteins from the cell.
What is mgcl2 in lysis buffer?
MgCl2 is a major component of lysis buffer in DNA extraction. Here MgCl2 breaks the cell membrane with the help of Tris. After the lysis of the cell membrane, DNase can easily attack DNA and can break it. MgCl2 binds with DNA and protect it from DNase activity.
Why is Mg2+ added in cell lysis?
Mg2+ then bind to DNA and protect it against DNase proteins that are now in direct contact with the DNA. DNAse, as a protein in itself, is spared a degradation activity and can also be precipitated, if it is the target protein. Mg2+ also mimic the ionic interactions that might have occurred during and/or post lysis.
What is glycerol in lysis buffer?
you can also add 5-10% glycerol to your lysis buffer as this helps protein solubility/stability. proteins are generally temperature sensitive and will aggregate if kept too warm or too cold.