How do you determine Colligative properties

Victoria Simmons
Published Apr 13, 2026
You determine colligative properties by making measurements. The vapor pressure lowering ΔP relative to that of pure solvent A is ΔP=PA−P . To determine the boiling point, you use an apparatus like the one shown below. You heat the liquid and measure the temperature when it boils.
How do you find colligative properties?
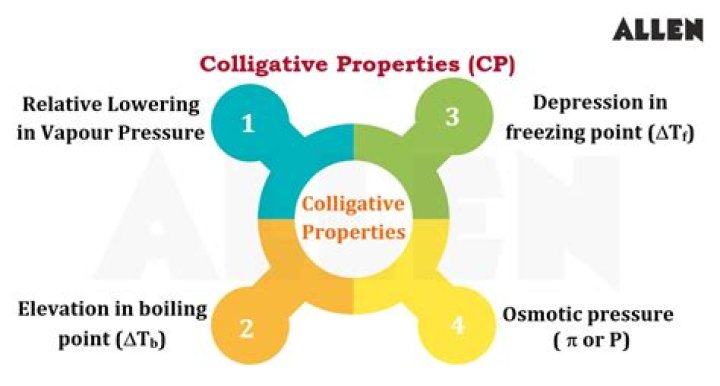
There are a few solution properties, however, that depend only upon the total concentration of solute species, regardless of their identities. These colligative properties include vapor pressure lowering, boiling point elevation, freezing point depression, and osmotic pressure.
What does colligative properties depend on?
By definition, one of the properties of a solution is a colligative property if it depends only on the ratio of the number of particles of solute and solvent in the solution, not the identity of the solute.
What determines the colligative properties of a solution?
Colligative Properties. Colligative properties of solutions are properties that depend upon the concentration of solute molecules or ions, but not upon the identity of the solute. Colligative properties include vapor pressure lowering, boiling point elevation, freezing point depression, and osmotic pressure.What are colligative properties how they are classified?
Colligative properties include lowering of vapour pressure, elevation of the boiling point, depression of the freezing point, and osmotic pressure. As will be defined in this module, this small set of properties is of fundamental significance to many natural phenomena and technical applications.
Which of the following are colligative properties?
Osmotic pressure is directly proportional to molarity and molarity is dependent on the number of solute particles and is independent on the nature of the solute particle. Hence osmotic pressure is a colligative property.
What is colligative property class 12?
Colligative properties are properties of a solution which depend only on the number of particles like ions or molecules of the solute in a definite amount of the solvent but not on the nature of the solute.
Do colligative properties depend on nature of solvent?
The colligative property of solution depend on the nature of solvent, as the solutions properties are similar to those of solvent, but do not depend on the nature of solute.What do you mean by colligative properties which colligative property is used to determine molar mass of polymer and why?
Osmotic pressure is the colligative property used to determine the molecular masses of polymer.
Which of the following colligative property can be determined at room temperature?The osmotic pressure method has an advantage because the pressure measurement is around room temperature.
Article first time published onDo colligative properties depend on number of particles?
Colligative properties depend only on the number of dissolved particles in solution and not on their identity. … Both solutions have the same freezing point, boiling point, vapor pressure, and osmotic pressure because those colligative properties of a solution only depend on the number of dissolved particles.
What are the limitations of colligative properties?
- When the solution freezes, the pure solvent alone must separate out.
- There must be no chemical reaction between solute and solvent. …
- The dissolved substance must not dissociate or associate appreciably.
What is a Colligative property give two examples?
Examples of colligative properties include vapor pressure lowering, freezing point depression, osmotic pressure, and boiling point elevation. … For example, adding alcohol (a volatile liquid) to water lowers the freezing point below that ordinarily seen for either pure alcohol or pure water.
What is Colligative property give example?
Colligative properties ⇨ Properties of solutions which depend on the number of solute particles but not on their nature. Examples of colligative properties are: Vapour Pressure lowering of a solution. Boiling Point elevation.
What are the four types of colligative properties?
There are four colligative properties: vapor pressure lowering, boiling point ele- vation, freezing point depression, and osmotic pressure.
How many colligative properties are there?
There are four colligative properties we will look at, which are: vapor pressure. boiling point. freezing point.
Which of the following Colligative property is most useful to determine?
The most important use of colligative properties in the laboratory is for determining the molecular mass of an unknown non-volative substance.
What does Delta TF represent?
∆Tf =i Kf m (electrolytes) Kf is the molal freezing point depression constant of the solvent (1.86 °C/m for water). m = molality = moles of solute per kilogram of solvent. Experiment.
Which Colligative property is widely used to determine molar masses of polymers ?`?
Measurement of osmotic pressure method is preferred for the determination of molar masses of macromolecules such as proteins and polymers.
How do you find molar mass from Colligative properties?
Use the freeing point depression (ΔTf) to calculate the molality of the solution. Then use the molality equation to calculate the moles of solute. Then divide the grams of solute by the moles to determine the molar mass.
How is colligative property related to molar mass?
For a given solute-solvent mass ratio, all colligative properties are inversely proportional to solute molar mass.
Which colligative property is most suitable to determine molar mass of proteins Why?
Osmotic pressure is the best property to determine the molar mass of biomolecules because it contains concentration in terms of molarity.
Which of the following is not a colligative property?
Elevation of Boiling Point. Depression of Freezing Point. Osmosis and Osmotic Pressure. Hence, the correct option is D.
Which of the following property does not depend upon the number of solute particles only?
Which of the following property does not depend upon the number of solute particles only? It is Relative lowering in vapour pressure which is a colligative property and not lowering in vapour pressure.
What is PS in Raoult's Law?
ps= vapour pressure of pure solution. Relative lowering of vapour pressure = p-ps/p. Raoult’s Law- According to Raoult’s law, “The relative lowering of vapour pressure of solvent is equal to the mole fraction of the solute.”
Which colligative property is the best method for determination of molar masses of macromolecules?
Osmotic pressure measurement is preferred for molar mass determination because : a even in dilute solution the osmotic pressure values are appreciably high and can be measured accurately.
What are Colligative properties name the colligative property which is widely used to determine the molecular mass of macromolecules?
Answer : (i) The colligative property which is used to find the molecular mass of macromolecules is osmotic pressure. By the above formula, we can calculate the molecular mass of macromolecules.
Which colligative property is best to determine molecular mass of macromolecules?
Osmotic pressure is preferred colligative property to determine molecular mass of macromolecules. – It can be measured with precision even in dilute solutions.
What do Colligative properties depend on Brainly?
Colligative properties depend on the number of solute particles in the solution.
Why depression in freezing point is a colligative property?
The freezing point depression depends upon the molal concentration of the solute and does not depend upon the nature of the solute . It is therefore, a colligative property.
How osmotic pressure is a colligative property?
Solutions. … colligative property depend on the number of solute particles irrespective of their nature relative to the total number of particles present in the solution. Osmotic pressure depends upon the number of particles of solute, i.e., molarity of solute. Therefore, it is considered as colligative property.