How are ionic bonds formed

Rachel Hickman
Published Apr 10, 2026
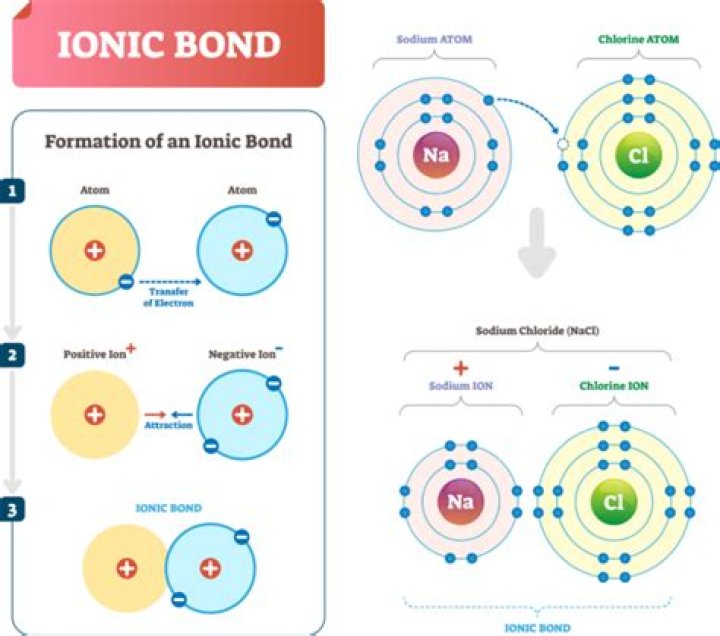
ionic bond, also called electrovalent bond, type of linkage formed from the electrostatic attraction between oppositely charged ions in a chemical compound. Such a bond forms when the valence (outermost) electrons of one atom are transferred permanently to another atom.
How are ionic bonds formed Quizizz?
Ionic Bonding | Chemical Bonds Quiz – Quizizz. Q. Ionic bonds form when ions share electrons. … Ionic bonds form only between atoms of nonmetals.
How are ionic crystals formed?
Ionic crystals are formed from ions through electrostatic attraction. One of the most well-known ionic crystals is table salt. Learn more about chemistry by exploring examples of chemical bonds.
How do ionic compounds form crystals?
The ions have a regular, repeating arrangement called an ionic lattice . The lattice is formed because the ions attract each other and form a regular pattern with oppositely charged ions next to each other. … This is why solid ionic compounds form crystals with regular shapes.Why do ionic bonds form between ions?
Formation of ionic bond An ionic bond can be formed after two or more atoms loss or gain electrons to form an ion. Ionic bonds occur between metals, losing electrons, and nonmetals, gaining electrons. Ions with opposite charges will attract one another creating an ionic bond.
How many steps are involved in the formation of an ionic bond *?
Three Steps For Creating An Ionic Bond.
What is ionic bonds quizlet?
Ionic bonds is a bond that forms when electrons is being transferred from one form to another form. … Ionic bonds form so that the outermost energy level of atoms are filled. Ion. an atom or group of atoms that bring out a positive or negative electric charge as a result of having lost or gained one or more electrons.
When an ionic compound is formed its charge is?
Ionic compounds are compounds composed of ions, charged particles that form when an atom (or group of atoms) gains or loses electrons. (A cation is a positively charged ion; an anion is a negatively charged ion.) Covalent or molecular compounds form when elements share electrons in a covalent bond to form molecules.Why do ionic compounds form complex crystal structures?
In order to minimize the potential energy of the system, ionic compounds take on the form of an extended three-dimensional array of alternating cations and anions. … This maximizes the attractive forces between the oppositely charges ions.
Do all ionic bonds make crystals?Crystals. Many compounds form molecules, but ionic compounds form crystals instead. A crystal consists of many alternating positive and negative ions bonded together in a matrix. Look at the crystal of sodium chloride (NaCl) in the Figure below.
Article first time published onHow are covalent crystals formed?
Covalent Bonding Network solids means the atoms form a network with each atom connected to four other atoms. This bonding in effect creates one large molecule that is tightly packed together. This characteristic defines covalent crystals and makes them structurally different from molecular crystals.
How could you identify that two elements have formed an ionic bond?
Two elements will form an ionic compound when a metal and a nonmetal will bond. One element will lose its electron and the other will gain its electron. A metal is a cation and a nonmetal is an anion.
How do covalent bonds form?
A covalent bond forms when the difference between the electronegativities of two atoms is too small for an electron transfer to occur to form ions. Shared electrons located in the space between the two nuclei are called bonding electrons. The bonded pair is the “glue” that holds the atoms together in molecular units.
How do ionic bonds form and how are the ions arranged in an ionic compound?
The ions in a solid ionic compound are not randomly arranged. Instead, they have a regular, repeating arrangement called an ionic lattice . The lattice is formed because the ions attract each other and form a regular pattern with oppositely charged ions next to each other.
Why do ionic bonds form quizlet?
ionic bonds form when electrons are transferred from one atom to another atom. ions of different elements can combine by forming ionic bonds . positive ions & negative ions form when atom s lose or gain electrons. … Atoms ,non-metal of elements tend to lose electrons when they form bonds.
How are ionic bonds formed between metals and nonmetals?
Ionic bonds form when a nonmetal and a metal exchange electrons, while covalent bonds form when electrons are shared between two nonmetals. An ionic bond is a type of chemical bond formed through an electrostatic attraction between two oppositely charged ions.
Which molecule is form by ionic bonds quizlet?
In an ionic bond, the atoms are bound together by the attraction between oppositely-charged ions. For example, sodium and chloride form an ionic bond, to make NaCl, or table salt. In a covalent bond, the atoms are bound by shared electrons.
Why do ionic bonds form between metals and nonmetals quizlet?
an ionic bond forms when the valence electrons of a metal atom is/are completely lost to a nonmetal atom. the metal loses an electron and becomes positively charged, while the nonmetal gains an electron and becomes negatively charged.
Why is the formation of ionic compounds exothermic?
Have you ever wondered why the formation of ionic compounds is exothermic? … The extra energy from the ions is released as heat when ionic bonds form. When more heat is released from a reaction than is needed for it to happen, the reaction is exothermic.
Which pair will form an ionic bond?
Ionic bonds usually occur between metal and nonmetal ions. For example, sodium (Na), a metal, and chloride (Cl), a nonmetal, form an ionic bond to make NaCl. In a covalent bond, the atoms bond by sharing electrons. Covalent bonds usually occur between nonmetals.
What is an ionic bond explain the formation of NaCl and caf2?
Two fluorine atoms, each one accepts one electron to become two uninegative fluoride ions (F- ) thereby attaining the stable configuration of Neon. 5. These three ions combine to form an ionic crystal in which they are held together by electrostatic attractive force.
What forces hold ionic crystals together?
Ionic Crystals. Ions in ionic crystals are bound together by electrostatic attraction.
Why do ionic bonds have high melting points?
Ionic compounds are held together by strong electrostatic forces of attraction between oppositely charged ions. These compounds have high melting points and high boiling points because of the large amounts of energy needed to break the many strong bonds. … These bonds between atoms are strong.
What keeps ions together in ionic compounds?
Ionic Bonds Oppositely charged particles attract each other. This attractive force is often referred to as an electrostatic force. An ionic bond is the electrostatic force that holds ions together in an ionic compound.
Are ionic compounds always crystalline?
Ionic compounds usually form crystalline structures when solid. … Ionic compounds typically have high melting and boiling points, and are hard and brittle. As solids they are almost always electrically insulating, but when melted or dissolved they become highly conductive, because the ions are mobilized.
How are metallic crystals formed?
Atoms in metals lose electrons to form cations. Delocalized electrons surround the ions. Metallic bonds (electrostatic interactions between the ions and the electron cloud) hold the metallic solid together. … This is due to the tightly packed crystal lattice of the metallic structure.
What type of structure does an ionic compound have?
Ionic compounds have regular structures, called giant ionic lattices . In a giant ionic lattice, there are strong electrostatic forces of attraction acting in all directions between the oppositely charged ions. The structure and bonding of ionic compounds explain their properties .
What type of bond is formed by the compounds?
Covalent BondsIonic BondsOccurs between:Two non-metalsOne metal and one non-metal
Why do covalent compounds do not form crystal lattice?
Covalent (or network) solids are extended-lattice compounds, in which each atom is covalently bonded to its nearest neighbors. Because there are no delocalized electrons, covalent solids do not conduct electricity. … Generally, covalent solids are insoluble due to the difficulty of solvating very large molecules.
What type of crystalline solid is formed from metal atoms?
Ionic crystals are composed of alternating positive and negative ions. Metallic crystals consist of metal cations surrounded by a “sea” of mobile valence electrons. Covalent crystals are composed of atoms which are covalently bonded to one another.
How is ionic bond formed on what factors it depends?
Ionic bonding is a chemical bond between a positive ion and negative ion. Ionization energy, electronegativity, and lattice energy are the factors that influence the formation of ionic bonds of elements.