Does insulin phosphorylate

Rachel Hickman
Published Mar 28, 2026
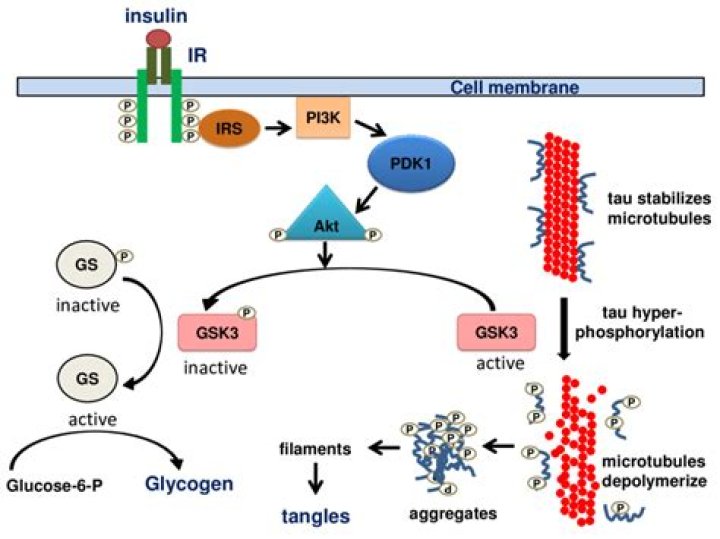
Insulin interacts with α subunit and induces a conformational change, leading to autophosphorylation of β subunits at tyrosine residues. It activates IR that recruits and phosphorylates scaffolds proteins to regulate mitogenic and metabolic functions (Belfiore et al., 2017).
Does insulin lead to phosphorylate or dephosphorylate?
Insulin induces the dephosphorylation of eIF2B at the site phosphorylated by GSK3, thereby stimulating the synthesis of protein from amino acids.
How does insulin affect potassium?
Insulin shifts potassium into cells by stimulating the activity of Na+-H+ antiporter on cell membrane, promoting the entry of sodium into cells, which leads to activation of the Na+-K+ ATPase, causing an electrogenic influx of potassium.
Does insulin phosphorylate GSK3?
Insulin was found to induce a marked phosphorylation of both GSK3 isoforms in WT control mice (Figure 2B).Is insulin a glucose transporter?
Insulin increases glucose uptake into fat and muscle cells through the regulated trafficking of vesicles that contain glucose transporter type 4 (GLUT4).
Does glucagon phosphorylate or dephosphorylate?
Also, the general rule that insulin promotes dephosphorylation, while glucagon promotes phosphorylation via cAMP/protein kinase A, will hold up very well throughout the course.
Does insulin phosphorylate or dephosphorylate HSL?
Insulin not only differentially regulates HSL isoform transcription but also post-transcriptionally affects HSL phosphorylation by stimulating PKA and endothelin (ET-1), and controls its expression indirectly via regulating the activity of growth hormone (GH).
What is GSK3 inhibitor?
In summary, GSK3 inhibitors effectively lower blood glucose in rodent models of type 2 diabetes, their effects occurring primarily through an increase in hepatic glycogen synthesis and a decrease in hepatic gluconeogenesis.What happens if GSK3 is inhibited?
GS inhibition by GSK-3β leads to a decrease in glycogen synthesis in the liver and muscles, along with increased blood glucose or hyperglycemia. This is why GSK-3β is associated with the pathogenesis and progression of many diseases, such as diabetes, obesity, cancer, and Alzheimer’s disease.
How is GSK3 activated?In addition to D2 receptors, GSK3 is also involved in signaling by other G protein-coupled receptors (Figure 4). Stimulation of serotonin (5HT) 5HT1A receptors increases the inhibitory serine-phosphorylation of GSK3, whereas 5HT2A receptor stimulation decreases it to activate GSK3 (Li et al., 2004).
Article first time published onDoes insulin increase potassium levels?
Insulin also increases the permiability of many cells to potassium, magnesium and phosphate ions. The effect on potassium is clinically important. Insulin activates sodium-potassium ATPases in many cells, causing a flux of potassium into cells.
Does insulin deplete potassium?
Insulin helps move potassium into cells. This can lead to hypokalemia, or low potassium levels in the blood. Untreated, the condition can cause problems with breathing and heart function—and even death.
Does insulin reduce potassium levels?
IV insulin leads to a dose-dependent decline in serum potassium levels [16]. A combination of IV insulin dose of 10 units plus 25 g of dextrose reliably lowers the serum potassium level by 1 mEq/L (mmol/L) within 10–20 minutes and the effect lasts about 4-6 hours [17, 18].
How is insulin transported?
Insulin is transported into the CNS by a saturable receptor-mediated process that is proposed to be dependent on the insulin receptor. Transport of insulin into the brain is dependent on numerous factors including diet, glycemia, a diabetic state and notably, obesity.
Do insulin receptors activate membrane glucose transport?
Insulin stimulates the translocation of a pool of Glut4 to the plasma membrane, through a process of targeted exocytosis.
How is insulin transported through the cell membrane?
The insulin circulates through the blood stream until it binds to an insulin receptor embedded in the cell membrane of a muscle, fat, or brain cell. Once the insulin binds to the receptor, phosphate groups are added to the intracellular domain of the receptor.
How does insulin inhibit HSL?
Insulin inhibits lipolytic activity by decreasing the phosphorylation and thus activity of HSL.
What stimulates HSL?
HSL is activated when the body needs to mobilize energy stores, and so responds positively to catecholamines, ACTH. It is inhibited by insulin. Previously, glucagon was thought to activate HSL, however the removal of insulin’s inhibitory effects (“cutting the brakes”) is the source of activation.
What is active HSL?
Active-HSL is a dietary formula to support a faster metabolism and relieve stress using a blend of natural ingredients. … It is based on the principle of healthy weight loss, which means its ingredients will help in weight loss without affecting any other body function.
Does insulin phosphorylate PFK2?
Insulin activates a protein phosphatase which dephosphorylates the PFK-2 complex and causes favored PFK-2 activity. PFK-2 then increases production of F-2,6-P2. As this product allosterically activates PFK-1, it activates glycolysis and inhibits gluconeogenesis. In contrast, glucagon increases FBPase-2 activity.
Does glucagon stimulate PFK2?
PFK2 is regulated by the hormones glucagon in the liver, epinephrine in muscle and by insulin. Both glucagon and epinephrine stimulate adenylate cyclase and cAMP-dependent protein kinase (PKA) in liver.
How does glucagon and insulin regulate pyruvate kinase?
Glucagon stimulates gluconeogenesis in part by decreasing the rate of phosphoenolpyruvate disposal by pyruvate kinase. Glucagon, via cyclic AMP (cAMP) and the cAMP-dependent protein kinase, enhances phosphorylation of pyruvate kinase, phosphofructokinase, and fructose-1,6-bisphosphatase.
What does GSK-3 do to glycogen synthase?
Glycogen synthase kinase 3 (GSK3) was identified over 20 years ago as a protein kinase that phosphorylated and inhibited glycogen synthase [1], the enzyme that catalyzes the transfer of glucose from UDPG to glycogen.
What is the molecular function of GSK-3?
Glycogen synthase kinase 3 (GSK3) is a serine/threonine kinase that was originally identified as a regulator of cell metabolism but has a variety of roles in cellular function including cell survival, proliferation, neural development, and neurotransmission [1, 2].
Is mTOR a kinase?
mTOR is a member of the phosphatidylinositol 3-kinase-related kinase family of protein kinases. … As a core component of mTORC2, mTOR also functions as a tyrosine protein kinase that promotes the activation of insulin receptors and insulin-like growth factor 1 receptors.
What hormone controls glycogen?
TermDefinitionglucagona hormone that tells cells in your liver and muscles to convert glycogen into glucose and release it into your blood so your cells can use it for energypancreasan organ in your abdomen that makes and releases insulin and glucagon
What is the process of Glycogenesis?
Glycogenesis is the process of glycogen synthesis, in which glucose molecules are added to chains of glycogen for storage. This process is activated during rest periods following the Cori cycle, in the liver, and also activated by insulin in response to high glucose levels.
What is the function of glycogen synthase kinase?
Glycogen synthase kinase 3β (GSK3β) is a serine/threonine kinase that is involved in various biological processes, including such as metabolism, cell cycle, transcription regulation, vesicular transport, neuronal function, oncogenesis, and development (Jope & Johnson, 2004).
How is GSK-3 inactivated?
GSK-3 activity is inhibited through phosphorylation of serine 21 in GSK-3α and serine 9 in GSK-3β. … Protein kinase A physically associates with, phosphorylates, and inactivates both isoforms of GSK-3.
What deactivates glycogen kinase?
Part of the mechanism by which insulin stimulates glycogen synthesis may involve phosphorylation and activation of Akt, serine phosphorylation and deactivation of glycogen synthase kinase-3 (GSK-3), leading to dephosphorylation and activation of glycogen synthase.
Does phosphorylation activate glycogen synthase?
Glycogen synthase activity is regulated by phosphorylation and allosterically activated by glucose 6-phosphate. … Glucose 6-phosphate increases both enzyme activity and substrate affinity.