Does calcium have a +2 ion

Robert Spencer
Published Apr 23, 2026
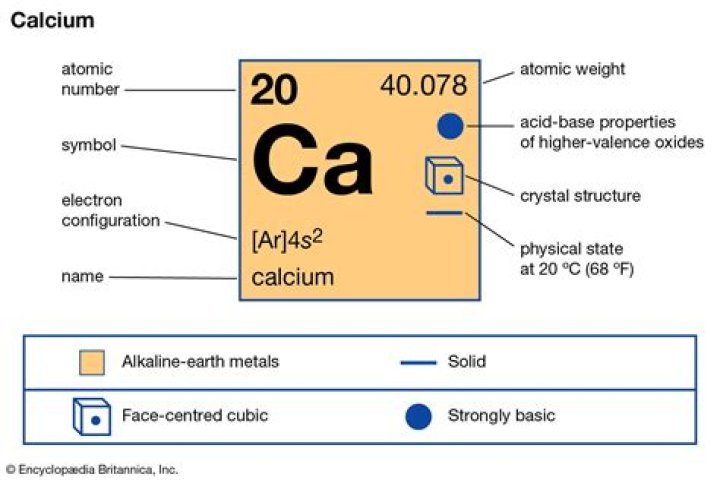
Calcium(2+) is a calcium cation, a divalent metal cation and a monoatomic dication. It has a role as a human metabolite and a cofactor.
What does a charge of +2 mean?
If this particular particle now has a positive 2 charge (+2), that means it either gained 2 protons, or lost 2 electrons. If we are talking about chemical elements, then it would mean that the element (particle) has LOST TWO ELECTRONS because electrons are outside the nucleus whereas protons are in the nucleus.
What is the difference between Ca and Ca2+?
2 Answers. For a) the neutral Ca atom has a larger radius than the Ca2+ ion. This is because the loss of electrons reduces the shielding effect between the electrons and the nucleus, so the attraction of the nucleus for the electrons is greater, reducing the radius.
What ion has a charge of +2?
Thus, a magnesium atom will form a cation with two fewer electrons than protons and a charge of 2+. The symbol for the ion is Mg2+, and it is called a magnesium ion.What does Ca 2 mean?
The calcium ion is also known as Ca2+, meaning it has two less electrons than protons and is not stable in nature. As a pure element, it’s a grey metal.
What does 2+ mean in Chem?
Ba2+ means that you have a Barium ion with two positive charges. so the 2 counts the positive charges and the + is not the symbol to add something, the + means only a positive charge.
What is the charge of calcium?
Calcium (Ca2+) has a positive charge. Carbonate (CO32-) has a negative charge. Because of these opposite charges, these ions naturally pair with one another (similar to two magnets). When this happens with these two particular ions, the result is calcium carbonate.
What does the subscript 2+ mean in Ca 2?
If a charge is present, it’s indicated in superscript, with a sign (+/-) and a number if more than one charge is present. For example, calcium ions have two positive charges so are written Ca2+.What happens when an element has a 2+ charge?
The atomic number for an element is the number of protons in the nuclei of their atoms. In a neutral atom, there are equal numbers of protons and electrons. Since the atom has gained a charge of 2- , it now has two more electrons than protons.
Why is Mg +2?Element NameMagnesiumElement SymbolMgAtomic Number12
Article first time published onWhy does the magnesium ion have a 2+ charge?
Magnesium has two electrons in its outer shell as it is in group 2 of the periodic table, which it loses, so its outer shell is full (2,8) and it has a charge of +2, as it still has 12 protons (positive charges) and now has only 10 electrons (negative charges).
What does calcium ion do?
Calcium ions (Ca2+) contribute to the physiology and biochemistry of organisms’ cells. They play an important role in signal transduction pathways, where they act as a second messenger, in neurotransmitter release from neurons, in contraction of all muscle cell types, and in fertilization.
Why does Ca2+ have a smaller radius than Ca?
Cations are smaller than their parent atoms, and so the Ca2+ ion is smaller than the Ca atom. Because Ca is below Mg in group 2A of the periodic table, Ca2+ is larger than Mg2+.
How is a calcium ion formed from calcium atom?
Calcium, Ca Calcium is also in Group 2. It has two electrons in its outer shell. When these electrons are lost, a calcium ion, Ca 2+, is formed.
Why is the calcium ion Ca2+ more stable than the calcium atom Ca?
Why is the calcium ion (Ca2+) more stable than the calcium atom (Ca)? The calcium atom has 2 electrons in a valence level above a stable noble gas configuration-If the atom can change into an ion identical to that of a noble gas, it can achieve the stability of the noble gas.
What is the charge of K?
ElementProtonsNet ChargePotassium atom190Potassium ion19+1Sulfur atom160Sulfur ion16−2
What is the charge of MG?
A magnesium atom must lose two electrons to have the same number electrons as an atom of the previous noble gas, neon. Thus, a magnesium atom will form a cation with two fewer electrons than protons and a charge of 2+. The symbol for the ion is Mg2+, and it is called a magnesium ion.
What does 2 mean on an element?
Helium is the element that is atomic number 2 on the periodic table. Each helium atom has 2 protons in its atomic nucleus. The atomic weight of the element is 4.0026.
What does the big 2 in 2nh3 mean?
An ammonia molecules contains one nitrogen and three hydrogen atoms. Leaving out the “1”, this becomes NH. The leading 2 indicates that you took this from a place where two of these molecules are present.
What two particles determine the mass number?
This explains why the mass of an atom is primarily determined by the mass of the protons and neutrons in the nucleus.
Why do some elements have 2 ion charges?
Many transition metals cannot lose enough electrons to attain a noble-gas electron configuration. In addition, the majority of transition metals are capable of adopting ions with different charges. … Because most transition metals have two valence electrons, the charge of 2+ is a very common one for their ions.
Why does iron have a +2 charge?
In the first compound, the iron ion has a 2+ charge because there are two Cl− ions in the formula (1− charge on each chloride ion). In the second compound, the iron ion has a 3+ charge, as indicated by the three Cl− ions in the formula. These are two different compounds that need two different names.
What elements gain 2 electrons?
For example, oxygen atoms gain two electrons to form O2- ions. These have the same electron configuration as the noble gas neon.
Is charge written as 2+ or +2?
When writing the chemical formula for an ion, its net charge is written in superscript immediately after the chemical structure for the molecule/atom. The net charge is written with the magnitude before the sign; that is, a doubly charged cation is indicated as 2+ instead of +2.
What are the 2 purposes of subscripts in chemical formulas?
The letter or letters that represent an element are called its atomic symbol. The numbers appearing as subscripts in the chemical formula indicate the number of atoms of the element immediately before the subscript. If no subscript appears, one atom of that element is present.
What is the charge on the common monatomic ion of lithium?
ABlithium ionLi+sodium ionNa+potassium ionK+rubidium ionRb+
Why is Mg positively charged?
. It has two electrons in 3s orbital and on losing these two electrons, its one shell will be removed and it will become more stable by coming close to the nucleus. Thus, whenever it loses these electrons, it becomes a positively charged ion.
Why does the Na ion have a charge of 1+?
A sodium atom has one electron in its outer shell. … A sodium atom can lose its outer electron. It will still have 11 positive protons but only 10 negative electrons. So, the overall charge is +1.
What is the electron configuration of Mg 2+?
Magnesium Cation Mg2+ Mg2+ has an electronic configuration of 1s² 2s² 2p^6, i.e., has a total number of 10 electrons similar to that of a Noble gas [Ne] instead of Mg 12 as its total number of electrons and configuration of 1s² 2s² 2p6 3s².
Is calcium ion an anion or cation?
FamilyElementIon NamePotassiumPotassium cationIIABerylliumBeryllium cationMagnesiumMagnesium cationCalciumCalcium cation
How many calcium ions are there in calcium fluoride?
CaF2, Calcium Fluoride, Fluorite The unit cell has 4 Ca2+ ions and 8 F1- ions. The material is transparent in the visible spectral region, and shows electronic optical adsorption in the ultra violet and lattice optical absorption in the infrared.