Does c2 only have pi bonds

Christopher Lucas
Published Mar 24, 2026
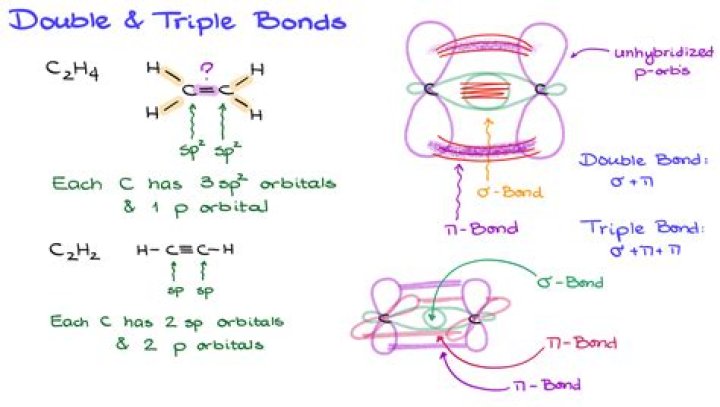
C2 molecules have been found in vapour state. Their double bonds are made of two pi bonds because four electrons need to be accomodated in each bond. So this is against the rule that there must be a sigma bond before a pi bond is formed in the double bond.
Why does C2 only have pi bonds?
Their double bonds are made of two $\pi $ bonds because four electrons need to be accommodated in each bond. In bond formation only valence electrons or outermost electrons participate. Hence, in ${{C}_{2}}$ molecules only 2$\pi $ are present. So, the correct answer is “Option C”.
Does B2 have only pi bond?
In B2+, no π bond exists, only two σ bonds.
How many bonds does C2 have?
The bonding components in C2 consist of two weakly bonding σ bonds and two electron-sharing π bonds.What bond does C2 form?
Rather, C2 is best described as having a traditional covalent σ bond with the electrons in the remaining orbitals of the two carbon atoms antiferromagnetically coupled.
Which orbital is the LUMO in C2?
According to Molecular orbital theory which of the following is correct (1) LUMO level for C2 molecule is 02px orbital (2) In C2 molecules both the bonds are a bonds (3) In C22-ion there is one o and two a bonds (4) All the above are correct.
How does C2 have 2 pi bonds?
This question already has answers here: C2 molecules have been found in vapour state. Their double bonds are made of two pi bonds because four electrons need to be accomodated in each bond. So this is against the rule that there must be a sigma bond before a pi bond is formed in the double bond.
How many sigma bonds does C2 2 have?
two σ bonds and one π – bond.Is C2 possible?
c2 molecule exist in space as gas, but under normal environment it cannot exist as 4 electron bonding with another 4 electron (quadruple bond) is not stable due to large repulsion between electron (same charge repel) and is very unstable.
Which compound is C2?Namesshow SMILESPropertiesChemical formulaC2Molar mass24.022 g·mol−1
Article first time published onHow many pi bonds are present in C2?
1σ and 3π
Why B2 and C2 have no sigma bonds?
In B2 you only have 2*3= 6 valence electrons. The first four will fill up the sigma(s) and sigma*(s). That produces bond order zero, so there is no sigma bond from that. The two remaining electrons will go into the two pi(p) orbitals, because they are lower than the sigma(s).
Which of the following species contains only pi bond?
The molecular orbital electronic configuration of C2 is: σ 1s2,σ∗ 1s2,σ 2s2,σ∗ 2s2,π 2p2x =π 2p2y. Thus C2 has only pi bonds according to molecular orbital theory.
Is C2 a quadruple bond?
Since it is highly untenable to assume that there is a chemical bond between two nonadjacent atoms of a linear molecule such as C3, we are led to the conclusion that the unexpected behavior of the interference energy of the “fourth” bond cannot be attributed to a new type of chemical bond and that there is no quadruple …
Does C2 have a quadruple bond?
Carbon is well known to form single, double, and triple C−C bonds in compounds. There is a recent report (2012) that carbon forms a quadruple bond in diatomic carbon, C2.
Why C2 molecule does not have sigma bond?
Therefore, the bond order of ${C_2}$ molecule is two. This means there should exist a double bond between the two carbons in a ${C_2}$ molecule. … These 4 electrons are in the pi orbitals and thus the two bonds in the ${C_2}$ molecule will be pi bonds only and no sigma bond.
Does C2 exist in Vapour phase?
In vapour phase, C2 molecule is diamagnetic.
Is C2 paramagnetic?
B2 has two unpaired electrons, therefore, B2 is paramagnetic. C2 has no unpaired electron, therefore, C2 is diamagnetic.
Is C2 a bond order?
The bond order of C2 molecule is 2. Bond order is the number of bonds between the two atoms which are participating in bond formation. C2 molecule does not exist in general.
Which has a stronger bond C2 or C2?
The bond order results show that the C2- has the highest number of valence electrons and highest bond energy. Therefore, C2- supposedly has the shortest bond length. C2+ has lesser bond energy than C2.
What is the electron configuration for C2?
MoleculeElectron ConfigurationBond OrderC2(σ2s)2(σ∗2s)2(π2py,π2pz)42N2(σ2s)2(σ∗2s)2(π2py,π2pz)4(σ2px)23
Does Dicarbon exist?
Carbon, the sixth most abundant element in the universe, has been known since ancient times. … Three naturally occurring allotropes of carbon are known to exist: amorphous, graphite and diamond. Amorphous carbon is formed when a material containing carbon is burned without enough oxygen for it to burn completely.
Why C2 does not exist in nature?
cannot exist as a stable molecule. Carbon hybridises most of the times to have 4-lone electrons ready to bond. In , the maximum number of bonds that can be formed is three (one sigma and two pi). This leaves one unpaired electron, making a free radical.
What is C2 in chemistry?
Diatomic carbon (C2) is historically an elusive chemical species.
Where are pi bonds found?
A pi bond (π bond) is a bond formed by the overlap of orbitals in a side-by-side fashion with the electron density concentrated above and below the plane of the nuclei of the bonding atoms.
Is C2 a compound or element?
C2 is considered a molecule but not a compound. Molecules are made of two or more atoms bonded together.
Does carbon exist as C2 or C?
Diatomic carbon or dicarbon (C2) exists only at very high temperatures in carbon vapour produced, for example, in electric arcs, in comets, stellar atmospheres and the interstellar medium, and in blue hydrocarbon flames.
What is C Chem?
Carbon (from Latin: carbo “coal”) is a chemical element with the symbol C and atomic number 6. It is nonmetallic and tetravalent—making four electrons available to form covalent chemical bonds. It belongs to group 14 of the periodic table.
Is C2 molecule diamagnetic?
C2 is diamagnetic because all of its electrons are paired.
What is the structure of C2 molecule?
Shaik has suggested that a quadruplebond exists in diatomic carbon . Molecular Orbital Theory shows that there are two sets of paired electrons in a degenerate pi bonding set of orbitals. This gives a bond order of 2, meaning that there should exist a double bond between the two carbons in a C2 molecule.
Can pi bonds exist without sigma bond?
A pi bond can exist between two atoms that do not have a net sigma-bonding effect between them. In certain metal complexes, pi interactions between a metal atom and alkyne and alkene pi antibonding orbitals form pi-bonds.