Do compounds have a charge

Christopher Lucas
Published Mar 24, 2026
A compound, by definition, has an overall net charge of zero. You can put a compound in a bottle. If every molecule had a charge, positive or negative, then every molecule would repel every other molecule.
What is the charge of a compound?
The atoms in chemical compounds are held together by attractive electrostatic interactions known as chemical bonds. Ionic compounds contain positively and negatively charged ions in a ratio that results in an overall charge of zero.
Do all compounds have electrical charge?
Terms in this set (12) all compounds that have an electrical charge of O;that is they are neutral. indicates the electric charge of an atom when electrons are lost, gained, or shared during chemical bond formation. the strong electron donors are on the left, and the strong electron acceptors are on the right.
Do solid compounds have charges?
Most cations and anions can combine to form neutral, solid compounds that are usually known as salts. The net charge of an ionic compound must be zero.Do ionic compounds have charges?
Ionic compounds are compounds composed of ions, charged particles that form when an atom (or group of atoms) gains or loses electrons. (A cation is a positively charged ion; an anion is a negatively charged ion.) … Molecular compounds are electrically neutral.
What is the charge of a solid?
Peter Dis1G wrote: So can I understand it as when solid is left a single element it has no charge.
Do compounds have to have a neutral charge?
Because the ionic compound must be electrically neutral, it must have the same number of positive and negative charges.
Are all compounds neutral?
Note: Even all compounds are neutral, some of them exhibit polarity. Because of the difference in electron affinity of the constituent atoms, the shared electrons are pulled towards the atom with high affinity to electrons. This happens in covalent bonds generally.Why do ionic compounds have a neutral charge?
Ionic compounds are electrically neutral because the charges of the cations and anions that make up the compound cancel each other out. … A chemical bond formed between two ions with opposite charges. Ionic bonds form when one atom gives up one or more electrons to another atom.
Are covalent compounds metal?Covalent BondsIonic BondsOccurs between:Two non-metalsOne metal and one non-metal
Article first time published onIs NaCl ionic or covalent?
Ionic bonds usually occur between metal and nonmetal ions. For example, sodium (Na), a metal, and chloride (Cl), a nonmetal, form an ionic bond to make NaCl. In a covalent bond, the atoms bond by sharing electrons.
Do covalent compounds have a charge?
Covalent compounds generally have low boiling and melting points, and are found in all three physical states at room temperature. Covalent compounds do not conduct electricity; this is because covalent compounds do not have charged particles capable of transporting electrons.
What is the total charge of all compounds?
Since an ionic compound consists of equal number of positive and negative ions, the overall charge of an ionic compound is zero.
How do you know the charge of an ionic compound?
- the number of charges on an ion formed by a metal is equal to the group number of the metal.
- the number of charges on an ion formed by a non-metal is equal to the group number minus eight.
- hydrogen forms H + ions.
What is the charge of the electron?
From the definition of the ampere, the electron itself has a negative charge of 1.602176634 × 10−19 coulomb.
What are ionic charges?
ionic charge The electrical charge of an ion, created by the gain (negative charge) or loss (positive charge) of one or more electrons from an atom or group of atoms.
How do you find the charge of a covalent compound?
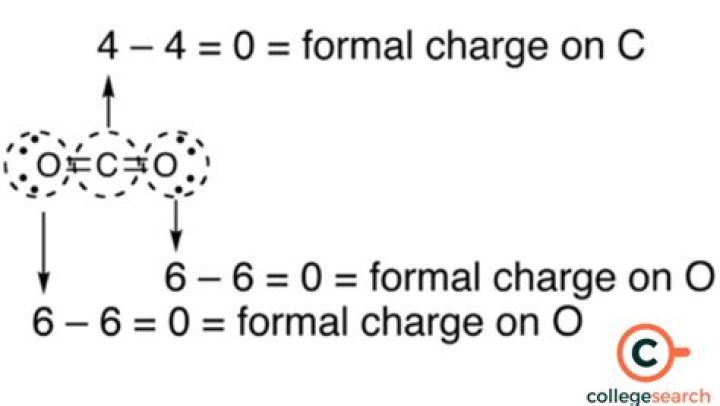
In a molecule, you can find the net charge of each atom. The formula is: number of valence electrons – number of non-bonding electrons – (number of sharing electrons * 1/2). This is called the formal charge. Then you find the sum of the formal charges (which will be the formal charge of the molecule).
Why are compounds always neutral?
Ionic compounds are electrically neutral because they form ionic bonds between positively and negatively charged atoms.
Why do elements carry a charge?
The charge on an atom is related to its valence electrons or oxidation state. An atom of an element is most stable when its outer electron shell is completely filled or half-filled. The most common charges are based on maximum stability for the atom.
Are ionic compounds neutral or do they have a charge?
Ionic compounds are neutral compounds made up of positively charged ions called cations and negatively charged ions called anions. For binary ionic compounds (ionic compounds that contain only two types of elements), the compounds are named by writing the name of the cation first followed by the name of the anion.
Are all surfaces negatively charged?
Surface charge practically always appears on the particle surface when it is placed into a fluid. Most fluids contain ions, positive (cations) and negative (anions). These ions interact with the object surface. … Dissociation of the surface chemical group is another possible mechanism leading to surface charge.
What surfaces are negatively charged?
- Titanium Dioxide.
- Graphene Oxide.
- Nanoparticles.
- Adsorption.
- Protein.
- Charged Surface.
- Functional Group.
- Hydrophobic.
Why do minerals have charges when they are submerged in water?
After all, as scientists have already known for a long time, a mineral surface acquires a charge if it is dampened with static water because some ions dissolve better than others. … Its surface has a negative charge in neutral and alkaline water.
Why are cations positively charged?
A cation has more protons than electrons, consequently giving it a net positive charge. For a cation to form, one or more electrons must be lost, typically pulled away by atoms with a stronger affinity for them.
Are covalent compounds neutral?
In covalent bonds, atoms share electrons, whereas in ionic bonds atoms transfer electrons. The reaction components of covalent bonds are electrically neutral, whereas for ionic bonds they are both charged. This explains why sodium chloride (salt) conducts electricity when dissolved — its components are charged.
What does all compounds are neutral mean?
Key Points. The overall ionic formula for a compound must be electrically neutral, meaning it has no charge.
Which is not ionic compounds?
Answer : Non-ionic compounds are those that have covalent bonds. Hydrochloric acid (HCl) and carbon tetrachloride (CCl4) form covalent due to sharing of a pair of electrons between two atoms.
How ionic compounds are formed?
An ionic bond is formed by the complete transfer of some electrons from one atom to another. The atom losing one or more electrons becomes a cation—a positively charged ion. The atom gaining one or more electron becomes an anion—a negatively charged ion.
What is a negative charged ion called?
The atom that has lost an electron becomes a positively charged ion (called a cation), while the atom that picks up the extra electron becomes a negatively charged ion (called an anion).
Do ionic compounds conduct electricity?
Ionic compounds do not conduct electricity in solid state as the ions are not free to move. Ionic compounds conduct as liquids or when in solution as the ions are free to move.
What are the difference between ionic and covalent compounds?
An ionic bond essentially donates an electron to the other atom participating in the bond, while electrons in a covalent bond are shared equally between the atoms. The only pure covalent bonds occur between identical atoms. … Ionic bonds form between a metal and a nonmetal. Covalent bonds form between two nonmetals.