Can PF5 dissolve in water

Mia Morrison
Published Feb 22, 2026
NamesDensity5.527 kg/m3 (g/L)Melting point−93.78 °C (−136.80 °F; 179.37 K)Boiling point−84.6 °C (−120.3 °F; 188.6 K)Solubility in waterhydrolysis
Why is PF5 not polar?
The molecular geometry of Phosphorous Pentafluoride (PF5 ) is trigonal bipyramidal. … PF5 has symmetric charge distribution of Fluorine atoms around the central atom Phosphorous. As charge distribution is equal and there is no net dipole moment therefore, this molecule is nonpolar.
What is PF5 used for?
Phosphorus Pentafluoride is a colorless gas with an unpleasant odor. It is used as a source of Phosphorus in semi- conductors and as a catalyst in ionic polymerization.
Is PF5 a dipole?
Due to their different three-dimensional structures, some molecules with polar bonds have a net dipole moment (HCl, CH2O, NH3, and CHCl3), indicated in blue, whereas others do not because the bond dipole moments cancel (BCl3, CCl4, PF5, and SF6).Is PF5 a base?
PF5 and PF4OH are both Lewis acids and form relatively stable Lewis acid-base complexes with H2O and EC through their oxygen lone pairs with complexation enthalpies of -41.8, -38.9, and -20.1 kJ mol-1 for PF5∙H2O, PF5∙EC, and PF4OH∙EC, respectively.
Is o3 polar or nonpolar?
Ozone has two resonance forms, each with a double bond, and each with a non bonding pair of electrons on the central atom, thereby ‘squishing’ the shape of the molecule into a ‘bent’ molecular geometry. The bent geometry results in a net dipole moment, and thus, ozone has a dipole moment, and thus, is polar.
What type of solid is PF5?
Phosphorus pentafluoride has 5 regions of electron density around the central phosphorus atom (5 bonds, no lone pairs). The resulting shape is a trigonal bipyramidal in which three fluorine atoms occupy equatorial and two occupy axial positions.
Does PCl5 have polar bonds?
PCl5 has a trigonal bipyramidal shape. The P-Cl bonds are polar, but they are symmetrically arranged around the central phosphorus atom resulting in no electrical dipole. So, the molecule is non-polar.Is PF5 a planar molecule?
Structure of phosphorus pentafluoride, PF5. There are three “equatorial” electron domains which lie in the same plane as the central P atom, forming a trigonal planar structure.
Is Cl2 polar or nonpolar?Is Cl2 polar or nonpolar? The electron charge is identical on both atoms. It is, therefore, a non-polar molecule.
Article first time published onWhat is the hybridization of PF5?
The hybridization is sp3d hybridization and phosphorous atom forms five sp3d hybrid orbitals. Five hybrid orbitals will be used to form bonds with five fluorine atoms.
What is BrCl5?
Bromine Pentachloride BrCl5 Molecular Weight — EndMemo.
What is the molar mass of PF5?
A sample of phosphorus pentafluoride (PF5; molar mass = 125.97 g/mol) is known to contain 0.244 moles of fluoride atoms.
Does PF5 follow octet rule?
And hence Pf5 does not obey octet rule.
Why does PF5 exist but not PH5?
Answer: PH5 is formed by the overlap of d orbitals with Sp3d hybridisation. F is more electronegative so Sp3d hybridisation is possible but H has lesser electronegativity and can not form sp3d hybridization and PH5 cannot be formed.
Does PF5 exist in solid state?
Let me correct your question: it is and not the substance that in solid state shows ionic structure. This happens because the molecular form and ionic form of phosphorus pentachloride have close values of standard Gibbs free energy. Thus, both forms can appear together in chemical equilibrium.
Is PF5 a Hypervalent?
Abstract: Some typical hypervalent molecules, SF4, PF5, and ClF3, as well as precursors SF (4SА state) and SF2 (3B1 state), are studied by means of the breathing-orbital valence bond (BOVB) method, chosen for its capability of combining com- pactness with accuracy of energetics.
How is PCl5 possible?
PCl5 forms five bonds by using the d-orbitals to “expand the octet” and have more “places” to put bonding pairs of electrons. NCl5 does not exist because there are no d-orbitals in the second energy level. Therefore there is no way to arrange five pairs of bonding electrons around a nitrogen atom.
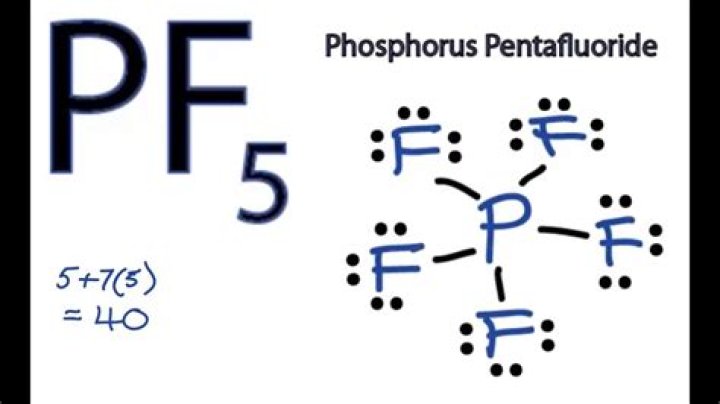
How many valence electrons does PF5?
In the Lewis structure for PF5 there are a total of 40 valence electrons. Five pairs will be used in the chemical bonds between the P and F.
Does O3 have dipole?
1b) ozone is a polar molecule (dipole moment = 0.5337 D).
Is O3 asymmetrical?
The distribution of electrons across the molecule is uneven – since the middle oxygen atom has to share electrons with two other atoms, but the other atoms only have to share electrons with one other atom. This results in the central atom having ‘less share’ of the electron density compared to the others.
What is the electronegativity of O3?
In ozone (O3) molecules, the two O−O bonds are nonpolar (there is no electronegativity difference between atoms of the same element).
Why is PCl5 soluble in water?
NamesSolubility in waterreactsSolubilitysoluble in CS2, chlorocarbons, benzeneVapor pressure1.11 kPa (80 °C) 4.58 kPa (100 °C)Structure
Why is PCl5 polar?
The difference between the electronegativity of Phosphorus and chlorine generates the polarity in the P-Cl bond. … But due to the symmetrical geometrical shape of PCl5 ie; trigonal bipyramidal, the polarity of the P-Cl bond gets canceled by each other and resulting in PCl5 an overall polar molecule.
How polar is chlorine?
Sodium has an electronegativity of 1.0, and chlorine has an electronegativity of 3.0. That’s an electronegativity difference of 2.0 (3.0 – 1.0), making the bond between the two atoms very, very polar.
What bond is Cl2?
In covalent bonds, like chlorine gas (Cl2), both atoms share and hold tightly onto each other’s electrons.
What shape is Cl2?
The molecular geometry of Cl2 is linear which is highly symmetrical and in straight lines. So, dipole generated in symmetrical shape can easily be canceled out. Also the equal number of lone pairs present on each chlorine atom in the Cl2 lewis structure.
What is the hybridization of the central atom of each of the following molecules PF5?
The hybridization of the central atom, P, is sp3d because it’s shape is trigonal bipyramidal.
Does PF5 have sp2 hybridization?
there are 5 sigma bonds in this compound. hence , the hybridization of this compound is sp3d . … 3 has sp2 hybridization.
What is the best hybridization of the P atom in PF5?
In PF5 phosphorus has 5 bond pair and 0 lone pair so steric no .is 5 . If steric no is 5 ,hybridization is sp3d. Añd shape of PF5 is Trigonal bi pyramydal (TBP).
Is NH4NO3 a covalent compound?
Ionic bond. NH4NO3 is a nitrate salt of the ammonium cation. Since ammonium is a cation and bonds with the anion nitrate, hence the compound is bonded by an ionic bond.