Can fungi grow in high pH

Sophia Edwards
Published Feb 16, 2026
As compared to other microorganisms, fungi are capable of growth and development over wide pH ranges. Several species, including plant pathogens, can actively modulate the pH of their environment by secreting acids or alkali (Landraud et al., 2013; Vylkova et al., 2017).
Do fungi like acidic or alkaline?
Most fungi inhabit mildly acidic environments, such as soil, plant, and animal surfaces.
How does pH affect mushroom growth?
As for the pH, which is the factor that we studied, the literature indicates that fungal growth increases with lower and that the peak of this increase is found at a pH of 4.5 (Rousk et al., 2009). Below that 4.5 threshold, fungal growth tends to significantly decrease.
Can fungi tolerate low pH?
Normally below pH 2.2 you may find very few fungi except Penicillium frequentans has been reported can tolerate up to pH 1.0. However in the alkaline range hardly any fungi are available to grow comfortably above pH 8.5.What pH level kills bacteria?
All microorganisms prefer a neutral pH for optimum growth, but they can grow in more acidic pH values. Most of them stop growing at a pH of 5.0.
Do fungi produce acid?
Fungi produce a wide variety of metabolites, including organic acid, vitamins, lipids, flavors, exopolysaccharides, amino acids, etc. Citric acid is the major organic acid produced by fungal fermentation and the second of all fermentation commodities following industrial ethanol.
What does the pH indicate?
pH is a measure of how acidic/basic water is. The range goes from 0 – 14, with 7 being neutral. pHs of less than 7 indicate acidity, whereas a pH of greater than 7 indicates a base. pH is really a measure of the relative amount of free hydrogen and hydroxyl ions in the water. … pH is reported in “logarithmic units”.
Does pH affect toenail fungus?
causes of onychomycosis – was shown in vitro to increase when the medium pH was increased from pH 4.5 to pH 7.5 and to decrease thereafter (Yazdanparast and Barton 2006). A higher toenail pH could favor the presence of fungal spores, which provide a reservoir for fungal infection.Do fungi require complex organic molecules to grow?
Like animals, fungi are heterotrophs; they use complex organic compounds as a source of carbon, rather than fix carbon dioxide from the atmosphere as do some bacteria and most plants. Like animals, they must obtain it from their diet. …
What pH level kills mold?However, what we need to emphasize here is that acetic acid is a slightly acidic compound with a pH of around 2.4, and that’s exactly what makes vinegar good at killing mold. Most mold species prefer to be in an acidic-neutral environment. In other words, they are most happy when the pH varies between 4 and 7.
Article first time published onIs alkaline water pH 9.5 good for you?
Healthline reveals that “normal drinking water generally has a neutral pH of 7; alkaline water typically has a pH of 8 or 9.” Results show that alkaline water is more beneficial by being able to effectively neutralize the acid in your body compared to other waters.
Does mold grow in alkaline conditions?
#6. Acid/alkaline requirements for yeast and mold growth in a wide range food products is quite broad, ranging from pH 2 to above pH 9.
How does pH affect mold growth?
Both yeasts and molds are able to grow in an acidic environment (pH less than 7). The pH range for yeast growth is 3.5 to 4.5 and for molds is 3.5 to 8.0. … Yeasts are unable to grow at a water activity of less than 0.9, and molds are unable to grow at a water activity below 0.8.
What conditions encourage bacteria multiply?
Bacteria can live in hotter and colder temperatures than humans, but they do best in a warm, moist, protein-rich environment that is pH neutral or slightly acidic.
Are all parasites pathogens?
The major difference between a parasite and a pathogen is that parasites are often a category of pathogen whereas pathogens are disease-causing organisms within the host. However, not all parasitic organisms are pathogens. Examples of pathogens include viruses, bacteria, and fungus.
What pH scale is vinegar?
Vinegar is mildly acidic with a pH of 2–3.
What pH is the most acidic?
The scale has values ranging from zero (the most acidic) to 14 (the most basic). As you can see from the pH scale above, pure water has a pH value of 7.
Is a pH of 7 acidic?
Acidic solutions (solutions with higher concentrations of H+ ions) are measured to have lower pH values than basic or alkaline solutions. … At 25 °C, solutions with a pH less than 7 are acidic, and solutions with a pH greater than 7 are basic. Solutions with a pH of 7 at this temperature are neutral (e.g. pure water).
Why is the pH of water 7?
pH is a measure of the amount of Hydrogen ions (H+) in a solution. … Even in pure water ions tend to form due to random processes (producing some H+ and OH- ions). The amount of H+ that is made in pure water is about equal to a pH of 7. That’s why 7 is neutral.
Which enzymes are produced by fungi?
IndustryFungal EnzymeAgricultureProtease, lipaseAnimal feedα-amylase, cellulase, lipase, phytase, protease, and xylanasePulp and paper productionCellulase, hemicellulase, lipase, and xylanaseWaste managementLipase
Which fungi produces gluconic acid?
The production of gluconic acid is mainly done in batch cultivation using several species belonging to the following fungal genera, Aspergillus, Penicillium, Fusarium, Mucor, and Gliocladium (Lockwood, 1975; Rosenberg et al., 1992; Petruccioli et al., 1994; Singh et al., 2001).
Which fungi produce oxalic acid?
Oxalic acid produced by an isolate of the mycorrhizal fungus Paxillus involutus (Batsch.
What nutrients do fungi need to grow?
Chemical elements such as phosphorus, sulfur, potassium, magnesium, and small quantities of iron, zinc, manganese, and copper are needed by most fungi for vigorous growth; elements such as calcium, molybdenum, and gallium are required by at least some species.
What conditions do fungi need to grow?
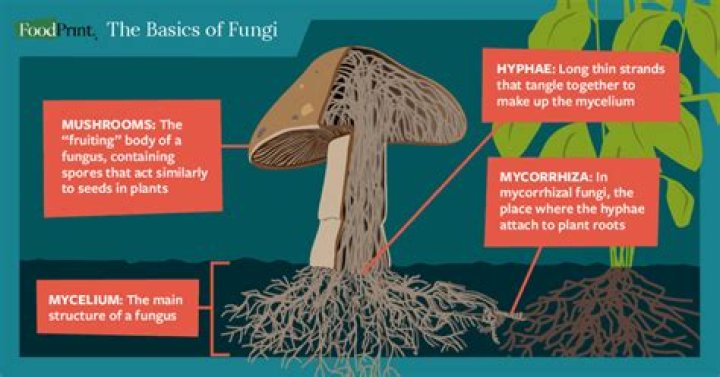
Like us, fungi can only live and grow if they have food, water and oxygen (O2) from the air – but fungi don’t chew food, drink water or breathe air. Instead, fungi grow as masses of narrow branched threads called hyphae.
Do fungi need sunlight?
Light: Fungi can only grow in the dark. For the most part, light does not play a role in how well fungi grow.
Can you use salicylic acid on toenail fungus?
Salicylic acid is used for a number of different skin conditions caused by thickened, hard skin, such as warts, psoriasis, scaly skin conditions and some nail infections. It is a keratolytic, which means that it works by softening the outer layer of your skin allowing it to loosen and shed.
Does ocean water help toenail fungus?
Finally, while it would be lovely to write off a seaside vacation as a medical expense, Lucy is sorry to inform you that, according to Dr. Larson, salt water has no medicinal properties when it comes to treating toenail fungus.
Do probiotics help with nail fungus?
Studies are continuing in regard to toenail fungus which is the same fungus responsible for “athlete’s foot” infections. Taking a proper Probiotic for a 2-3 month period will help “reset” your gut/immune system and allow your own body to fight the fungal infection without the use of drugs or topical medications.
What pH is a lemon?
Lemon juice has a pH between 2 and 3, which means it’s 10,000–100,000 times more acidic than water (1, 2, 3). A food’s pH is a measure of its acidity. The pH of lemon juice falls between 2 and 3, meaning it is acidic.
Is lemon water alkaline?
Lemon juice is acidic, with a pH of 3, while water has a pH of around 7, which is neutral. This means it is neither acidic nor alkaline.
Is alkaline water bad for kidneys?
There are no hard facts either way. But for most healthy individuals, drinking alkaline water is probably not harmful. If you have chronic kidney disease or are taking a medication that affects your kidney function, elements in alkaline water could possibly have negative side effects on the kidneys.