Can double bonds be chiral

Rachel Hickman
Published Mar 05, 2026
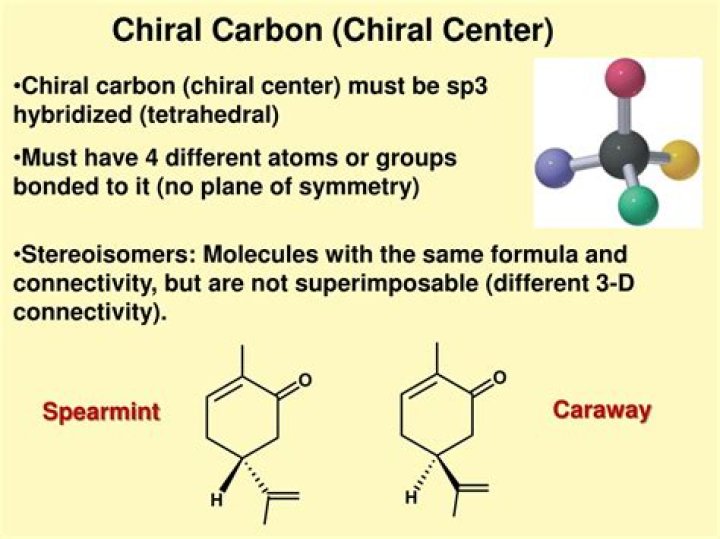
Chiral molecules usually contain at least one carbon atom with four nonidentical substituents. … Neither will carbons on double or triple bonds be chiral centers because they can’t have bonds to four different groups.
Are alkenes chiral functional groups?
Each can be superimposed on its own mirror image, and neither is chiral (also, note the lack of a chiral center!) However, they both have the same molecular formula and the same bonding connectivity, so by definition they are stereoisomers of each other.
How do you tell if an alkene can exist as a stereoisomer?
Stereoisomers are defined as molecules with the same structural formula but a different arrangement of the atoms in space. Owing to the restricted rotation around a C=C double bond it is possible for alkenes to exist as stereoisomers if there are two different groups attached to each carbon atom in the double bond.
Why are alkenes chiral?
An allene with two different substituents on each of the two carbon atoms will be chiral because there will no longer be any mirror planes.Are alkenes optically active?
Yes, allenes are optically active as long as they have different groups on each end of the cumulative double bonds. They are constrained in rotation, so they can be resolved. There are some naturally occurring allenes, some being optically active.
Is 2 propanol chiral?
2-propanol, unlike 2-butanol, is not a chiral molecule. Carbon #2 is bonded to two identical substituents (methyl groups), and so it is not a chiral center.
Why are double bonds not chiral?
Neither will carbons on double or triple bonds be chiral centers because they can’t have bonds to four different groups. When looking at a molecule, look for carbons that are substituted with four different groups. See, for example, if you can spot the two chiral centers in the molecule shown here.
Can planar molecules be chiral?
Planar chirality, also known as 2D chirality, is the special case of chirality for two dimensions. Most fundamentally, planar chirality is a mathematical term, finding use in chemistry, physics and related physical sciences, for example, in astronomy, optics and metamaterials.Can Allenes be chiral?
Cumulated dienes (allenes) also exhibit chirality without having asymmetric carbon atoms. A cumulated diene is a molecule that contains two double bonds on a single carbon. The central carbon atoms of allene are sp-hybridized.
Are enantiomers chiral?Chiral describes an atom with four different groups attached, while enantiomers describes the comparison between two stereoisomers. Enantiomers do have chiral centers in the molecules, but not all stereoisomers of a molecule are enantiomers of each other.
Article first time published onCan alkenes have enantiomers?
It has a chirality center and therefore can exist as two enantiomers: The question is which one forms in excess, or in other words what is the stereochemistry of this, and in general, for the electrophilic addition reactions of alkenes.
Are alkenes planar?
Alkenes, containing a carbon-carbon double bond, have the trigonal planar geometry as a primary feature. A portion of the molecule is flat and only 2-dimensional.
Why is Allene unstable?
Why is allene unstable? – Quora. An Allene has two π bonds. Interestingly the two π bonds are perpendicular to each other,due to which there is no overlapping of the vacant p orbitals,ultimately leading to no delocalization of electrons. This accounts for the instability of allenes.
What is alkenes general formula?
Alkenes are defined as either branched or unbranched hydrocarbons that possess at least one carbon–carbon double bond (CC) and have a general formula of CnH2n [1].
Can constitutional isomers have double bonds?
There are two ways in which isomers can differ. When they differ in the way the atoms are connected, they are called constitutional isomers. Butane and isobutane, for example, are constitutional isomers. … The carbon atoms in the C=C double bond in 2-butene, for example, are stereocenters.
For which double bonds are stereoisomers possible?
Alkenes, which are molecules containing carbon-carbon double bonds, have the possibility of having stereoisomers, just as ring systems do. This is because, unlike carbon-carbon single bonds, which are free to rotate, double bonds are fixed and rigid.
Which molecule can exist as stereoisomers?
Both compound A (1-bromo-1-chloropropene) and compound B ( 1-cyclobutyl-2-ethyl-3-methyl-1-butene) can exist as a pair of configurational stereoisomers (one is shown).
Why are alkenes optically inactive?
In the previous section, the addition of water to the achiral alkene produced a racemic mixture of two enantiomeric alcohols. They are produced in equal amounts so the mixture is optically inactive. … That intermediate does not have a plane of symmetry and therefore attack by water is not equal from the top and bottom.
Can double bonds be optically active?
The double-bonded (sp2) atom itself cannot be a chiral center, but one can have alkenes which are optically active at an sp3 center. For example, 3-methylpent-1-ene has an (R) and an (S) form due to a chiral center at C3.
Can alkenes show optical isomerism?
no, alkenes doesn’t show optical isomer due to presence of plane of symmetry.
Do alkenes have stereochemistry?
Alkenes, which are molecules containing carbon-carbon double bonds, have the possibility of having stereoisomers, just as ring systems do. … You can only have cis-trans stereochemistry in rings and on double bonds.
Can alkenes be diastereomers?
E/Z isomers of alkenes are “stereoisomers that are not enantiomers” too! So they are diastereomers. The double bond is the non-chiral source of the stereochemistry.
Is Z the same as CIS?
So Z resembles “cis” and E resembles “trans” . (Note: they are not necessarily the same and do not always correlate: see footnote for an example of a cis alkene which is E .
How do you determine Superimposability?
The most straightforward way to determine whether a given object is chiral is to draw or visualize the object’s mirror image and see if the two are identical (that is, superimposable). If the object contains an internal plane of symmetry then it must be achiral.
Is propanol chiral or achiral?
2-Propanol, an achiral molecule It is not a chirality center, because the methyl groups are not distinguishable. Each 2-propanol molecule is superimposable to its mirror image.
Are all chiral centers Stereocenters?
A stereocenter is any atom in a molecule for which exchanging two groups creates a different stereoisomer. All chiral centers are stereocenters, however, not all stereocenters are chiral centers as we will encounter examples of this in later chapters.
Are Allenes alkenes?
The properties of allenes are similar to those of alkenes, although the pure compounds often are difficult to prepare and are not indefinitely stable. Allenes undergo many of the usual double-bond reactions, being readily hydrogenated, adding bromine, and being oxidized with potassium permanganate solution.
Is allene a planar molecule?
(Hint: allene is not a planar molecule.)
Is Mycomycin chiral?
Mycomycin contains an allene, which is chiral, and the whol molecule has two optically active enantiomers. Originally Answered: A molecule may not have a chiral atom but still be chiral. On the other hand a molecule may have more than one chiral atoms but still be achiral.
What makes a molecule chiral?
Chiral molecules contain one or more chiral centers, which are almost always tetrahedral (sp3-hybridized) carbons with four different substituents. … A is not superimposable on its mirror image (B), thus by definition A is a chiral molecule.
How do you know if a molecule is chiral?
Test 1: Draw the mirror image of the molecule and see if the two molecules are the same or different. If they are different, then the molecule is chiral. If they are the same, then it is not chiral.