Can alkenes be enantiomers

Sophia Edwards
Published Mar 05, 2026
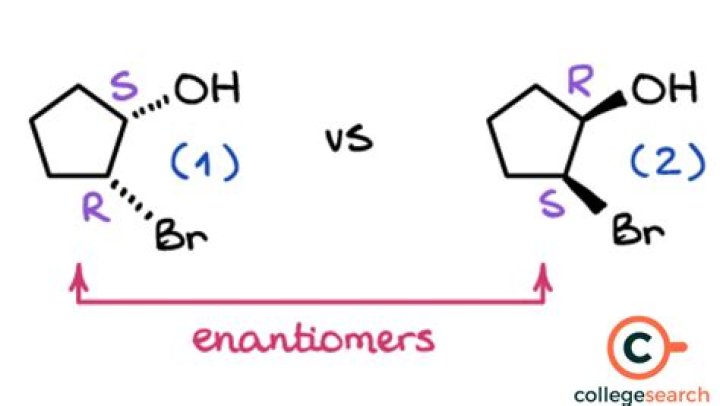
It has a chirality center and therefore can exist as two enantiomers: The question is which one forms in excess, or in other words what is the stereochemistry of this, and in general, for the electrophilic addition reactions of alkenes.
Can alkenes be stereoisomers?
Alkene stereochemistry. As we have already described, alkenes with two different substituents at each end of the C=C can exist as a pair of stereoisomers. The alkene can only exist as stereoisomers if R1 is not equal to R2 AND R3 is not equal to R4.
Are alkenes diastereomers?
E/Z isomers of alkenes are “stereoisomers that are not enantiomers” too! So they are diastereomers. The double bond is the non-chiral source of the stereochemistry.
Can alkenes be chiral?
Alkenes have no classical chirality, so generally, an external stereogenic center must be introduced. However, by locking the alkene into a conformation through the use of an achiral buckle allows for the creation of an inherently chiral alkene.Can any molecule have an enantiomer?
Any given molecule has its enantiomer; the two other molecules are its diastereomers.
Can alkenes have Stereocenters?
A stereocenter is an atom for which the interchange of two groups converts one stereoisomer into another. … An unambiguous system for describing stereoisomers of alkenes has been developed, in which strict rules are used to assign a priority to the substituents on each end of the double bond.
Are enantiomers chiral?
Chiral describes an atom with four different groups attached, while enantiomers describes the comparison between two stereoisomers. Enantiomers do have chiral centers in the molecules, but not all stereoisomers of a molecule are enantiomers of each other.
Are alkenes chiral functional groups?
Each can be superimposed on its own mirror image, and neither is chiral (also, note the lack of a chiral center!) However, they both have the same molecular formula and the same bonding connectivity, so by definition they are stereoisomers of each other.Are alkenes optically active?
Yes, allenes are optically active as long as they have different groups on each end of the cumulative double bonds. They are constrained in rotation, so they can be resolved. There are some naturally occurring allenes, some being optically active.
How can you tell the difference between enantiomers and diastereomers?EnantiomersDiastereomersEnantiomers are stereoisomers that are mirror images of each otherDiastereomers are stereoisomers that aren’t a mirror image of each otherHave identical physical properties except for the ability to rotate plane-polarised lightDistinct physical properties
Article first time published onCan enantiomers be diastereomers?
EnantiomersDiastereomersExample: Lactic acidExample: Tartaric acid
How do you make enantiomers?
Enantiomers are stereoisomers that are non-superimposable mirror images, meaning that one enantiomer will be the mirror image of the other enantiomer. In order to draw an enantiomer, you can determine the stereocenter, then swap the two groups attached to the stereocenter.
Are ring flips enantiomers?
Another interesting feature of this molecule is that a ring flip produces the enantiomer. Since both enantiomers have the same steric biases and are equal in energy, the ring flips between the enantiomers rapidly at room temperature.
Which pairs are enantiomers?
Enantiomers are pairs of compounds with exactly the same connectivity but opposite three-dimensional shapes. Enantiomers are not the same as each other; one enantiomer cannot be superimposed on the other. Enantiomers are mirror images of each other.
Which molecule has enantiomers?
Among molecules with the same connectivity: Molecules that are mirror images but non-superimposable are enantiomers.
Which compound can exist as enantiomers?
Cis-2-butene.
Are enantiomers isomers?
Enantiomers are two optical isomers (i.e. isomers that are reflections of each other). … Compounds that are enantiomers of each other have the same physical properties except for the direction in which they rotate polarized light and how they interact with different optical isomers of other compounds.
Are enantiomers always optically active?
Each enantiomer of a stereoisomeric pair is optically active and has an equal but opposite-in-sign specific rotation. Specific rotations are useful in that they are experimentally determined constants that characterize and identify pure enantiomers.
How do you identify enantiomers?
Bottom line for today: you can tell if molecules are enantiomers or diastereomers by looking at their (R,S) designations. Enantiomers are non-superimposable mirror images of each other. Pardon me while I push the Caps Lock button: ENANTIOMERS ALWAYS HAVE OPPOSITE R,S DESIGNATIONS.
Do alkenes have stereochemistry?
Alkenes, which are molecules containing carbon-carbon double bonds, have the possibility of having stereoisomers, just as ring systems do. … You can only have cis-trans stereochemistry in rings and on double bonds.
Can isomers have double bonds?
In order for geometric isomers to exist, there must be a rigid structure in the molecule to prevent free rotation around a bond. This occurs with a double bond or a ring. In addition, the two carbon atoms must each have two different groups attached in order for there to be geometric isomers.
Do isomers have the same gram formula mass?
Isomers have the same chemical formula. Therefore, the molecular weight or molar mass of the isomers will be the same.
Why alkenes do not show optical isomerism?
no, alkenes doesn’t show optical isomer due to presence of plane of symmetry. only geometrical isomerism they can show but even bonded cumulenes can show optical isomers but the co…
Why are alkenes optically inactive?
In the previous section, the addition of water to the achiral alkene produced a racemic mixture of two enantiomeric alcohols. They are produced in equal amounts so the mixture is optically inactive. … That intermediate does not have a plane of symmetry and therefore attack by water is not equal from the top and bottom.
Can double bonds be optically active?
The double-bonded (sp2) atom itself cannot be a chiral center, but one can have alkenes which are optically active at an sp3 center. For example, 3-methylpent-1-ene has an (R) and an (S) form due to a chiral center at C3.
What functional group is och3?
A methoxy group is the functional group consisting of a methyl group bound to oxygen. This alkoxy group has the formula O–CH3. On a benzene ring, the Hammett equation classifies a methoxy substituent at the para position as an electron-donating group, but as an electron-withdrawing group if at the meta position.
Are alkanes functional groups?
Alkane sequences form the inert framework of most organic compounds. For this reason, alkanes are not formally considered a functional group. When a hydrocarbon chain is connected as a substituent to a more fundamental structural unit, it is termed an alkyl group.
Are halogens a functional group?
Halogens are common functional groups for alkanes, and can be used in reactions to reduce an alkene into an alkane.
Are enantiomers the same molecule?
They are the same molecule and it is not correct to describe them as separate species or separate stereoisomers. They are a meso compound; a molecule with asymmetric carbon atoms whose mirror image is superimposable.
How do enantiomers differ?
Enantiomers differ only in their optical activity i.e. the direction in which they rotate plane polarized light. If an enantiomer rotates polarized light to the right or in a clockwise direction, it is said to be the (+) or the dextrorotatory isomer.
What's the difference between enantiomers and isomers?
Optical isomers are configurational isomers that differ in the 3D relationship of the substituents about one or more atoms. Enantiomers are optical isomers that are non-superimposable mirror images.